World Health & Population
Determinants of Gender Differences in Health among the Elderly in Latin America
Antonio J. Trujillo, Thomas A. Mroz, Claudia Piras, John A. Vernon and Gustavo Angeles
Abstract
This paper identifies the main gender differences in health and socio-economic characteristics of the elderly in four Latin American cities. Using locally weighted regressions as well as a flexible model specification that treats age non-parametrically, we investigate whether these unadjusted gender gaps in health are due to gender differences in the distribution of age and other explanatory variables. Interestingly, for all cities, the analyses show a gender gap in health in favour of males at each age. The gaps are larger when one uses functional impairment in mobility and personal self-care as indicators of an individual's health instead of self-reported health. Furthermore, controlling for demographic characteristics, baseline health and the availability of family support do little to change the disadvantage for women in measured health outcomes. Controlling for socio-economic variables does, however, reduce most of the gender differences in health.
I. Introduction
As the Latin American public becomes more aware of the economic, financial and social implications of the rapid demographic transition occurring in these economies, there is increased pressure to reform the network of health and social services that provide support to seniors. Available evidence suggests that the populations of Latin American countries are aging at a faster rate than those of many developed countries (Shrestha 2000). Regional trends track global ones, but some important differences are worth noting. The percentage of the population that is elderly in Latin America is projected to increase to 6.5% by the year 2010 and to 8.5% by 2020, growing much faster than the average for the rest of the globe. The expected growth in the proportion of the region's population over the age of 80, the "very elderly," also exceeds the global trend (Arriagada 2003). Rapid improvements in survival rates within this age group will have a tremendous impact on medical care expenditures, which in time will pressure governments to develop policies to meet this future demand.
The needs of these rapidly growing elderly populations, and in particular the health needs of the very elderly, may force these nations to develop new systems geared toward providing seniors with financial support, formal and informal social support, and healthcare. How the governments in Latin American countries decide to distribute the costs associated with their rapidly aging populations among different members of society will introduce fundamental economic and social challenges in the future. To effectively implement policy initiatives for the elderly, a necessary first step is to better understand the economic and non-economic determinants of health status among this group. To do this, special attention must be paid to how life histories and current demographic and socio-economic conditions affect the onset of illness and the speed at which health deteriorates among elderly males and females.
Policies aimed at improving the health status of the elderly have been justified on the grounds that they are part of a global strategy to reduce poverty and gender inequalities. Gender equity with respect to health and well-being among the elderly has become a crucial element in recent political reforms in Latin America (Saith and Harriss-White 1999). Assessment of well-being is measured with a set of social indicators; health measures such as mortality and morbidity rates are among the many dimensions used to construct more comprehensive indices usually referred as the functionings framework (Sen 1998).1 Under this approach, directly reducing genders gaps in health status among the elderly is one way to increase the overall well-being of a country's population.
It is a well-known fact that females live longer than males, but they also experience lower overall health status. Nathason's (1975) seminal piece and more recently the work by Knodel and Ofstedal (2003) review in detail the issues of gender differences in the aging experience. Although little empirical evidence exists for developing countries (see Strauss et al. 1993; Sen 1998; Buvinic et al. 2006), the results so far are consistent with the following paradoxical finding: Women in developing economies report worse health status and higher morbidity than males, in spite of longer life expectancies.
The size and composition of gender differentials in mortality, morbidity rates, self-reported health and medical care use vary over the course of life (Verbrugge 1985; Ross and Bird 1994 Valanis 1999; Arber and Cooper 1999). Statistics about morbidity and self-reported health status over the course of life indicate lower self-reported health status, higher morbidity from nonfatal chronic conditions, more frequent acute diseases and higher prevalence of mental problems and short-term disability for females, while males report a higher frequency of life-threatening conditions. The excess mortality in males seems to be the result of higher morbidity from life-threatening conditions rather than an overall excess morbidity. Women also report a higher use of heath services and therapeutic drugs than males.2 Lastly, in rankings of health problems that lead to medical care consumption and possible death, males and females are highly similar (Verbrugge 1985). However, the frequency, intensity and path to death of these illnesses differ substantially across gender.
A closer look at the gender gaps in health among the elderly indicates that gender differentials in mortality and morbidity diminish over the course of life (Verbrugge 1985). Later in life, females report lower self-reported health and greater functional limitations than males, due to disability. Females are also more prone to report nonfatal chronic conditions (e.g., hemorrhoids, constipation, dermatitis, thyroid conditions, anemia and arthritis) than males. Furthermore, females experience acute conditions and short-term infectious diseases more frequently than males. In contrast, elderly males are more likely to suffer from life-threatening chronic conditions, including coronary heart disease, malignant neoplasm, cerebrovascular disease, kidney disease and cirrhosis than females. Finally, in the later stages of life, as elderly males tend to increase their use of health services as their medical problems become more severe, the gender gap in the use of medical procedures and therapeutic drugs declines (Ross and Bird 1999; Valanis 1999).
Five different hypotheses have been postulated to explain the gender gaps in health (for an excellent review, see Verbrugge 1985): (1) biological risk of disease, (2) risk of disease and injury, (3) aspects of therapeutic actions, (4) health reporting behaviour and (5) prior healthcare use. The relevance of these factors may vary over the course of life. Males may undertake more risky behaviour than females (e.g., smoking and drinking) during adult life. Nevertheless, one might expect males would put their health at risk less often as they become older. Likewise, living arrangements may become more relevant later in life. Gender differences in education, earnings and occupation could influence one's health during adult life, and they may have a significant impact on health later in life. Biological differences that may favour females have been linked to childbearing years and are less important during later stages of life. Essentially, women have been found to exhibit a stronger resistance to illnesses during adult years since they need to be healthy to give birth (Ross and Bird 1999; Sen 1985.
In addition to these explanations, gender differences in health status and morbidity rates among the elderly could be due to the problem of selection. Since men have a greater mortality rate than females over the course of life, self-reported health status observed in the data for elderly males may be the residual result of the survival of the healthiest males. Accounting for gender differentials in mortality selection could explain the observed gender gap in health among the elderly.
In our analysis, we will develop a model that examines the following four central questions:
- What are the main differences in self-reported health and life-threatening and disabling conditions between elderly males and females in the four cities in our sample?
- What are the main differences between elderly males and females with respect to income, asset holdings and composition, education, marital status, present occupation, age of retirement and insurance status?
- Are gender differentials in health due to differences in age composition?
- What factors explain the patterns in gender differentials in self-reported health and functional disability among the elderly?
II. Methods and Data Description
A. Methods
Our first step will be to focus on describing the gender gaps in health among the elderly. This descriptive analysis is conducted using the SABE dataset for the following four cities: Buenos Aires (Argentina), Sao Paolo (Brazil), Santiago (Chile) and Ciudad de Mexico (Mexico). In the forthcoming sections, we use the country name to refer to each of these cities.
The analysis incorporates several dimensions of individual health, including cognitive evaluation, self-reported health status (SRHS), the prevalence of chronic and acute conditions, and functional health – Activities of Daily Living (ADL) and Instrumental Activities of Daily Living (IADL) indices – reported in the SABE project data.
SRHS has been widely recognized as a simple but very comprehensive indicator of an individual's health, and it is a good predictor of an individual's future mortality (Deeg and Kriegsman 2003). Additionally, self-reported physical functioning and the ability to perform personal care (ADL and IADL) have been widely used to study the health of populations in developed and developing societies (Strauss et al. 1993).
After describing elderly health, we will examine the demographic and socio-economic conditions facing the elderly. This analysis provides a clear picture of the main differences between male and female elderly seniors, with a focus on those factors that may affect their health.
Once an understanding of the gender differentials in health among the elderly has been provided, we will undertake an analysis to determine if these health gaps are the consequence of gender differences in age composition. The existence of differences in age composition implies, on one hand, that the unadjusted gender differences in health observed in the data may be the result of the higher proportion of females relative to males who reach old age, but who also report lower health. The average male's health may be the result of fewer healthier men who similarly reach old age. Therefore, the comparison of the unadjusted gender differences in health may be biased in favour of males because, on average, they will be younger than females.
Using cross sectional data,3 one could test this hypothesis by evaluating the effect of gender on health at each age. Essentially, after controlling for age, the remaining differences in health between males and females will not be the consequence of gender differences in age composition.
To test this hypothesis, the model we use will follow Grossman (1972), where health is treated as a stock of human capital. In particular, we implement two empirical approaches to explore the existence of gender differentials on health. First, we estimate a model where the age effects are unrestricted, and the female effect is assumed constant across all ages. Second, we estimate a more flexible model, where the female effect varies by age. In both cases, we undertake multiple regression analyses using sampling weights to account for having low numbers of elderly above 80 years of age.
The reduced-form equation we use to model the age-related effect of gender on health is the following:
Yi = a(60) + a(61)d(61)i + a(62)d(62)i+ …+a(98)d(98)i+b(F)Fi+ µi (1)
where Yi measures an individual's health. In our analysis, we use three different measures of health: SRHS, ADL and IADL. In addition, d61, d62, d63 … d98 are a vector of dummy variables for each age. In this case, we are assuming that the lowest age in the data is 60 and the highest is 98. Fi is a dummy variable for female. Lastly, µi represents a vector of the unobserved characteristics that influence an individual's health. One should notice that this model treats age non-parametrically and assumes a constant female effect for all ages. In particular, testing the hypothesis that the female coefficient is significant and negative will imply that elderly females report lower health, holding constant an individual's age. We estimate this model separately for each country in the sample.
Since this model may be too restrictive, in the sense that it assumes a constant effect of gender on health at each age, we also estimate the following more flexible model:
Yi = a(60) + a(61)d(61)i + a(62)d(62)i+ …+a(98)d(98)i+b(F)Fi+b(61)[Fid61i] + b(62)[Fid62i]+…+b(98)[Fid98i] +εi (2)
Since this model assumes a different gender effect on health at each age, testing the joint hypothesis that the female interaction effects are significantly different from zero will shed light on the gender effect on health after controlling for age.
Using the model presented in Equation 1, we also estimate a locally weighted regression using linear probability models.4 The idea behind this approach is to estimate a regression at each age using only a subset of the sample, weighting points close to each age more heavily than points farther away. In this way, we can reduce the influence of the distribution tails in the average calculation of health at each age. These estimated regressions are then used to predict a smoothed health value for each age. We undertake this analysis for each country. The results of this method are graphed for each measure of health used in the analysis.
Given the dependent variable in the analysis (health status), we had to choose between using a linear probability specification (ordinary least squares; OLS) or an alternative method to handle categorical dummies with more than two choices (e.g., ordinal logit, ordinal probit, count models, etc.). Since one of the main objectives of the research was to find out whether females at each age (instead of average health) report lower health than males, we decided to implement a non-parametric specification model in age. This implies including a dummy variable for each age and additional computational effort to estimate the average standard error of the age/sex variable. Given the complicated specification and, more importantly, the limited sample size in our database, it was not feasible to run models such as ordinal logit or probit. Yet, the model results are similar for the linear probability model, logit and probit specifications when one implements a standard parametric approach controlling for a single age variable and a dummy variable for gender.
Lastly, using the model presented in Equation 1, we attempt to explain the gender differences in health after controlling for relevant covariates in a sequential order. In our analysis, we divide the set of variables into four vectors: (1) a vector of demographic characteristics, (2) a vector of socio-economic conditions, (3) a vector of family support characteristics and (4) a vector of baseline health.5
For each country, we estimate the basic model (which only includes a separate dummy for each age and a dummy for female) and add to it each of the vectors separately. After including each vector of covariates, we test the individual hypothesis that b(F) is negative and statistically significant. By comparing the magnitude of the coefficients, we may evaluate which vector has the greatest effect on reducing the gender differentials in health status. This approach allows us to test if eliminating differences in current socio-economic conditions is sufficient to eliminate the gender gaps in health, and to test if these differences are more important in explaining the gender gaps in health than the other gender differences in demographic, family support and baseline health conditions.
B. Data Source, Relevance of the Data and Variable Description
The SABE database is a cross-sectional survey that collects information from 1999 and 2000 about the health status and health conditions of the elderly population in seven representative cities in Argentina, Barbados, Brazil, Chile, Cuba, Mexico and Uruguay.6 The survey instruments were consistent across countries, which gives a unique opportunity to compare the health status of the elderly in these cities. The survey includes representative samples of individuals older than 60 years of age living in urban areas. Furthermore, the sampling design accounts for potential problems of under-representation in the final sample of individuals over 80 years of age, as well as institutionalized individuals. The information included in the survey is similar to that provided by the Health and Retirement Survey (HRS) in the United States.
Table 1 describes all the dependent variables (SRHS, ADL and IADL), the independent variable (gender) and several of the control variables used in our statistical analyses.7 The vector of control variables includes the societal, cultural, environmental and biological risk factors affecting gender differentials in health outcomes. For convenience, the vector of control variables is categorized as demographic, socio-economic characteristics, family support characteristics and baseline health status.
| Table 1. Description of variables in the SABE database | |
| Variables | Description |
| Dependent variables | |
| SRHS | A question that includes the following options: excellent, very good, good, fair and poor |
| ADL/IADL scores | Variables to measure current ADL and IADL scores. This excludes any difficulties that an individual expects to last more than 3 months |
| Other health-related variables | |
| Cognitive evaluation | A vector of 10 different variables (e.g., memory at the present time, able to manage money, shopping alone, remember family events, etc.) |
| Existence of chronic and acute conditions | Different questions about the existence of hypertension, diabetes, cancer, lung disease, heart attack, coronary heart disease, angina or other heart problems, cerebral embolism, arthritis, rheumatism or osteoarthritis |
| Mental health | Variables to measure emotional, nervous or psychiatric problems in the last 12 months |
| Health risk factors | Smoking, drinking and eating behaviours |
| Independent variable | |
| Gender | Sex |
| Control variables | |
| Vector of current socio-economic characteristics | Level of schooling, illiteracy, age when started to work, current work status, age at retirement, type of occupation, total income from different sources (pension, family transfers, banking income, welfare subsidy), home ownership, list of household assets (e.g., refrigerator, washer, water heater, microwave, television, telephone, VCR, radio player, heating, air conditioning, fan) Availability of health insurance, includes the following categories: social security, private and public insurance |
| Vector of family support characteristics | Number of household members, number of brothers and sisters |
| Vector of baseline health status | Father or mother alive, father or mother's age at death, living in rural areas first 5 years of life, age when first diagnosed with cancer, past smoker, current smoker, family economic condition during the first 15 years of life, self-assessment of individual's health in the first 15 years of life, existence of any of the following illnesses during the first 15 years of life: kidney disease, hepatitis, measles, tuberculosis, rheumatic fever, asthma, bronchitis |
III. Results
In this section, we answer the four key questions we posed at the beginning of the paper. For the first two sections, we discuss the results for all the countries included in the analysis; however, we present only the results for Sao Paulo (Brazil). The rest of the findings are available from the authors upon request. For the last two sections, we show the results for all cities.
A. What Are the Main Differences in Health Between the Male and Female Elderly in These Four Cities?
In Brazil, for both genders, all measures of self-reported functional health status from the sample decline with age. Males have better indicators of functional mobility, ADL and IADL, than females across all age groups (see Table 2). As Hurd et al. (1999) have shown, this result may suggest that help from spouses obfuscates ADL limitations among males. Females have higher prevalence rates for all life-threatening conditions in all age groups, with the exception of chronic lung disease. Observe that the prevalence of some life-threatening conditions declines with age. This may indicate that individuals have died from the disease, but it could also be the result of the incidence of the disease by birth cohort. Diabetes displays this pattern across all age groups. The decline in diabetes prevalence between the age groups of 60 to 70 years and older than 85 years is more dramatic for males than for females: 17.4% to 8.1% and 19.4% to 15.1% for males and females, respectively.
| Table 2. Brazil - elderly health: gender comparison by age group, weighted summary statistics | ||||||||||||
| Health indicators | Males (881) | Females (1262) | ||||||||||
| (311) | (472) | (98) | (494) | (643) | (125) | |||||||
| Young old (60 < X < 70) |
Old (71 < 85) |
Old old (> 85) |
Young old (60 < X < 70) |
Old (71 < 85) |
Old old (>85) |
|||||||
| Mean | Std err | Mean | Std err | Mean | Std err | Mean | Std err | Mean | Std err | Mean | Std err | |
| Health functional status | ||||||||||||
| SRHS | 2.592 | (0.052) | 2.543 | (0.054) | 2.395 | (0.096) | 2.521 | (0.046) | 2.502 | (0.044) | 2.432 | (0.088) |
| ADL index | 7.923 | (0.146) | 7.132 | (0.145) | 5.530 | (0.271) | 6.723 | (0.137) | 5.930 | (0.133) | 4.364 | (0.252) |
| IADL index | 14.469 | (0.122) | 13.449 | (0.217) | 10.6 | (0.605) | 13.97 | (0.118) | 12.942 | (0.122) | 9.378 | (0.502) |
| Life-threatening conditions | ||||||||||||
| Hypertension | 0.489 | (0.029) | 0.516 | (0.028) | 0.392 | (0.055) | 0.546 | (0.025) | 0.593 | (0.024) | 0.588 | (0.043) |
| Diabetes | 0.174 | (0.023) | 0.173 | (0.023) | 0.081 | (0.028) | 0.194 | (0.018) | 0.182 | (0.017) | 0.151 | (0.033) |
| Cancer | 0.015 | (0.006) | 0.061 | (0.016) | 0.022 | (0.015) | 0.031 | (0.008) | 0.038 | (0.007) | 0.046 | (0.021) |
| Chronic lung disease | 0.132 | (0.018) | 0.161 | (0.020) | 0.184 | (0.051) | 0.107 | (0.013) | 0.108 | (0.015) | 0.106 | (0.035) |
| Heart disease | 0.173 | (0.017) | 0.262 | (0.024) | 0.215 | (0.048) | 0.158 | (0.018) | 0.223 | (0.016) | 0.285 | (0.045) |
| Stroke | 0.071 | (0.002) | 0.122 | (0.021) | 0.098 | (0.036) | 0.051 | (0.010) | 0.071 | (0.012) | 0.082 | (0.029) |
| Disabling conditions | ||||||||||||
| Arthritis, rheumatism or osteoarthritis | 0.196 | (0.023) | 0.223 | (0.022) | 0.267 | (0.046) | 0.390 | (0.019) | 0.423 | (0.021) | 0.420 | (0.049) |
| Fall in the last 12 months | 0.203 | (0.024) | 0.236 | (0.028) | 0.436 | (0.060) | 0.307 | (0.024) | 0.348 | (0.018) | 0.454 | (0.045) |
| Incontinence | 0.079 | (0.015) | 0.165 | (0.020) | 0.325 | (0.050) | 0.223 | (0.021) | 0.297 | (0.021) | 0.441 | (0.046) |
| Cognitive scores | 0.959 | (0.013) | 0.911 | (0.014) | 0.816 | (0.055) | 0.970 | (0.009) | 0.884 | (0.015) | 0.623 | (0.057) |
| Psychiatric problems | 0.124 | (0.017) | 0.127 | (0.018) | 0.119 | (0.045) | 0.198 | (0.019) | 0.160 | (0.017) | 0.074 | (0.026) |
| Anthropometry measures | ||||||||||||
| Height (cm) | 165.5 | (0.491) | 164.10 | (0.42) | 161.7 | (0.845) | 152.8 | (0.322) | 150.9 | (0.305) | 147.6 | (0.685) |
| Weight (kg) | 70.8 | (0.847) | 67.50 | (0.68) | 59.8 | (1.393) | 64.8 | (0.796) | 62.0 | (0.635) | 55.7 | (1.689) |
| Note. (1) SRHS was coded 5 = excellent; 4 = very good; 3 = good; 2 = fair; 1 = poor. (2) ADL is an indicator from 0 to 10 (0 = worst condition). (3) IADL is an indicator from 0 to 15 (0 = worst condition). (4) Each health condition refers to whether a doctor or nurse ever told the individual that he or she had the condition. A dummy indicator was constructed where 1= existence of the condition; 0 = otherwise. (5) Cognitive score represents the percentage of individuals with a score higher than 13 (that is, in good cognitive condition). | ||||||||||||
As shown in Table 2, the prevalence of physically disabling conditions (arthritis, rheumatism, osteoarthritis, falling and incontinence) increases with age for both males and females. The prevalence of physically disabling conditions is higher for females than males. For instance, while 26.7% of very elderly males report having arthritis, rheumatism or osteoarthritis, 42% of very elderly females report having these same conditions. Lastly, females are significantly more likely to suffer from incontinence than males – more than 10 percentage points higher than males. The cognitive capabilities of the elderly less than 85 years of age are very similar for both genders. However, female cognitive abilities decline drastically, and are comparatively lower than those of males, after 85 years of age.
In Argentina, males report better indicators of functional health status. Yet, males report higher instances of chronic lung disease, heart disease and stroke than females. As expected, for both genders, all measures of functional health status decline with age. The lower health functional indicators reported for females may reflect the severity of disabling conditions. For example, females are more likely to report arthritis, rheumatism or osteoarthritis, and falling in the last 12 months than males. Interestingly, both males and females in Argentina report the highest rates of arthritis, rheumatism or osteoarthritis among all countries in the study. The cognitive capabilities of the elderly less than 85 years of age are very similar for males and females. However, males' cognitive abilities decline considerably and are comparatively lower than those of females after 85 years of age (opposite of the pattern in Brazil).
In Chile, the gradient of health deterioration with age is less pronounced for males than females when one looks at health functional status measures. This faster decline in functional health status among females may be caused by the higher rates of hypertension, diabetes, cancer, heart disease and stroke. Chilean males and females report the lowest rates of diabetes in all age groups, compared to Brazil, Argentina and Mexico. As expected for both genders, the likelihood of all disabling conditions increases with age. As with previous results, disabling conditions are more prevalent for females. At any age, females are more likely to report a lower cognitive score than males.
Lastly, in Mexico, indicators of functional mobility are higher for males than for females in all age groups, despite the fact that the self-reported health status for males and females is very similar. Interestingly, ADL and IADL decline more precipitously with age in Mexico for both males and females than they do in other countries. There is no clear evidence to suggest that females are more likely than males to suffer from life-threatening conditions, which was the case in other countries. Females were also less likely to report diabetes, heart disease and stroke compared to males in all age groups, while males were less likely to report hypertension and cancer. Hypertension rates for males are significantly lower in Mexico than in other countries and slightly lower for females. Mexican males report the highest rates of diabetes in all age groups compared to Brazil, Argentina and Chile.
Regarding disabling conditions, as expected, females are more likely to report disabling condition than males. The cognitive scores declined with age, but the level of the cognitive scores for males and females older than 85 were the lowest in Mexico, when compared to the other countries: 0.560 for males and 0.493 for females.
In sum, females reported lower functional health status and higher prevalence of all disabling conditions than males. Cognitive functions showed a similar pattern. Saad (2002) and Bos and Bos (2005) have also reported an observed better health status of elderly males. A sample selection factor could provide one tentative explanation for these results. In particular, fewer males than females surpass the age of 60. Consequently, males who survive are, on average, healthier than surviving females. Regarding life-threatening conditions in all countries, females are more likely to report hypertension and cancer than males.
B. What Are the Main Differences between Male and Female Elderly in Income, Asset Level and Composition, Education, Marital Status, Present Occupation, Age of Retirement and Insurance Status?
In Brazil (see Table 3), males show a higher functional health status than females. However, females tend to have a similar number of children alive and similar family networks to males. This social network may have a positive effect on health. The better socio-economic conditions that males report may also indicate that males have more resources than females to protect their health. Specifically, males report higher rates of literacy and education and are more likely to be employed, to own their own home and to maintain private health insurance than females. Surprisingly, females report higher total wealth in all age groups, even though males report higher incomes. This may be the consequence of females living with other household members. These results are not consistent with the results reported by Hurd et al. (1999) in the case of the United States, where males show higher average wealth than females. In all age groups, males are more likely to report they are smokers, which may explain why males report higher instances of chronic lung disease.
| Table 3. Brazil - elderly characteristics of demographics, socio-economic status, family support and baseline health by age group, weighted summary statistics of selected variables | ||||||||||||
| Social indicators | Males (881) | Females (1262) | ||||||||||
| (311) | (472) | (98) | (494) | (643) | (125) | |||||||
| Young old (60 < X < 70) |
Old (71 < 85) |
Old old (> 85) |
Young old (60 < X < 70) |
Old (71 < 85) |
Old old (> 85) |
|||||||
| Mean | Std err | Mean | Std err | Mean | Std err | Mean | Std err | Mean | Std err | Mean | Std err | |
| Total children alive | 2.74 | (0.102) | 2.752 | (0.089) | 2.834 | (0.163) | 2.599 | (0.093) | 2.559 | (0.084) | 2.399 | (0.158) |
| Currently married | 0.821 | (0.023) | 0.771 | (0.022) | 0.537 | (0.049) | 0.508 | (0.024) | 0.321 | (0.018) | 0.052 | (0.021) |
| Number of marriages | 1.084 | (0.028) | 1.059 | (0.026) | 1.218 | (0.070) | 1.053 | (0.017) | 1.058 | (0.020) | 1.025 | (0.024) |
| Socio-economic conditions | ||||||||||||
| Literacy | 0.888 | (0.022) | 0.789 | (0.027) | 0.619 | (0.060) | 0.802 | (0.023) | 0.671 | (0.032) | 0.553 | (0.053) |
| Education | 0.866 | (0.026) | 0.765 | (0.028) | 0.568 | (0.053) | 0.823 | (0.022) | 0.709 | (0.026) | 0.612 | (0.054) |
| Years of education | 1.511 | (0.102) | 1.472 | (0.103) | 1.417 | (0.218) | 1.353 | (0.059) | 1.232 | (0.066) | 1.192 | (0.070) |
| Occupation | 1.780 | (0.055) | 1.687 | (0.051) | 1.605 | (0.102) | 1.558 | (0.062) | 1.433 | (0.056) | 1.346 | (0.112) |
| Retired | 0.497 | (0.031) | 0.238 | (0.024) | 0.013 | (0.008) | 0.211 | (0.018) | 0.096 | (0.012) | 0.021 | (0.013) |
| Age of retirement | 56.748 | (0.592) | 63.494 | (0.373) | 67.997 | (1.332) | 45.308 | (0.909) | 48.643 | (0.987) | 53.757 | (2.584) |
| Home ownership | 0.855 | (0.025) | 0.871 | (0.016) | 0.901 | (0.031) | 0.832 | (0.023) | 0.790 | (0.021) | 0.821 | (0.044) |
| Total wealth in dollars ($1 = 1.80 real) | 362.27 | (11.09) | 420.85 | (14.63) | 421.02 | (28.82) | 434.62 | (13.46) | 475.67 | (15.48) | 428.49 | (29.45) |
| Total income in dollars ($1 = 1.80 real) | 2132.36 | (244.66) | 2792.64 | (450.10) | 1299.42 | (225.66) | 1329.80 | (173.64) | 1405.07 | (156.98) | 1697.15 | (492.02) |
| Private health insurance | 0.058 | (0.014) | 0.073 | (0.014) | 0.114 | (0.044) | 0.044 | (0.009) | 0.048 | (0.011) | 0.011 | (0.007) |
| Family support | ||||||||||||
| Number of household members | 2.759 | (0.049) | 2.485 | (0.049) | 2.477 | (0.114) | 2.524 | (0.041) | 2.284 | (0.039) | 2.348 | (0.098) |
| Number of brothers and sisters | 3.553 | (0.072) | 3.155 | (0.077) | 2.115 | (0.156) | 3.356 | (0.061) | 2.833 | (0.061) | 1.919 | (0.101) |
| Baseline health status | ||||||||||||
| Living in rural areas first 5 years of life | 0.682 | (0.036) | 0.693 | (0.037) | 0.845 | (0.038) | 0.564 | (0.030) | 0.605 | (0.029) | 0.589 | (0.050) |
| Smoking | 0.268 | (0.024) | 0.160 | (0.019) | 0.112 | (0.031) | 0.120 | (0.020) | 0.103 | (0.014) | 0.060 | (0.025) |
| Initial family economic condition | 0.893 | (0.040) | 0.994 | (0.045) | 1.043 | (0.094) | 1.019 | (0.041) | 1.087 | (0.046) | 1.098 | (0.090) |
| Initial self-assessment of individual's health | 1.45 | (0.036) | 1.465 | (0.036) | 1.479 | (0.076) | 1.425 | (0.034) | 1.396 | (0.035) | 1.517 | (0.065) |
| Initial starvation | 0.248 | (0.026) | 0.168 | (0.018) | 0.161 | (0.037) | 0.211 | (0.019) | 0.154 | (0.016) | 0.112 | (0.029) |
| Note. (1) Total children included biological children, step-children and adopted children. (2) Education was coded 0 = no school; 1 = schooling. (3) Occupation was coded 1 = employee; 2 = manager; 3 = self-employed; 4 = other. (4) Years of education was coded 1 = elementary; 2 = secondary; 3 = technical; 4 = college. (5) Retired was coded 0 = retired; 1 = non-retired. (6) Home ownership was coded 1 = own; 0 = does not own. (7) Wealth is based on price index of assets in the household adjusted by household size. (8) Income includes working and non-working income adjusted by household size. (9) Initial family economic condition, self-assessment of individual's health and starvation refer to the first 15 years of life. (10) Smoking was coded 0 = not current smoker; 1 = current smoker. (11) Economic condition during the first 15 years of life was coded 0 = poor; 1 = average; 2 = above average. (12) Initial self-assessment of health refers to the first 15 years of life and was coded 0 = poor; 1 = good, 2 = excellent. | ||||||||||||
Like Brazil, in Argentina, males are more likely to be married at all ages than females, and they are also more likely to have a higher income level than females in all age groups. The higher functional health status of males compared to females, and the lower rate of life-threatening conditions like cancer and hypertension, may be a result of these differences. The literacy, education and years of education rates for this cohort are similar for both males and females in all age groups.
In Chile, total income is higher for males than for females in all age groups, which may explain the higher health functional status and lower rates of life-threatening conditions for males. There is not a large discrepancy between the education rates of males and females. In particular, for the elderly older than 85, a larger percentage of females have attended school than males. Males are more likely to be married than females in all age groups.
In Mexico, as in all countries, the rate of males currently married is higher in all age groups than for females. The level of total income is also higher for males, with the exception of the very elderly group. Males' literacy, education and years of education are higher than those of females. However, males' socio-economic advantages are not reflected in the distribution of life-threatening conditions.
In sum, in all countries included in the study, males are more likely to be married and show a more modest decline in the rate of marriage than females. Iacovou (2000) has reported that elderly European males are also more likely to be married than females, and there is little decline in the percentage of married males until age 80, when the proportion drops off more significantly. Males report higher education, are more likely to own their home, and report higher income than females.
C. Are Gender Differentials in Health Due to Differences in Age Composition?
In this section, we investigate whether the unadjusted gender differences in health reported in Section II. A are a consequence of gender differentials in mortality selection.
To explore this issue, we conduct two separate analyses: an unrestricted regression of age and gender on health, and a locally weighted regression. Table 4 summarizes the results for the first exploratory analysis. Figure 1 shows the results for the second analysis.
| Table 4. OLS unrestricted regression analysis of the effect of gender and age on the elderly's health; dependent variables - SRHS, ADL, IADL; weighted statistics | ||||||||
| OLS unrestricted regressions | Brazil | Argentina | Chile | Mexico | ||||
| I. Model includes a dummy for each age and assumes a constant female effect | ||||||||
| 1.a. SRHS | ||||||||
| Female coefficient | −0.058 | (0.041) | −0.259 | (0.068) | −0.245 | (0.078) | −0.159 | (0.052) |
| Fraction of negative age coefficients (min. age 61)a | 53.8% | 91.2% | 80.6% | 57.9% | ||||
| I.b. ADL | ||||||||
| Female coefficient | −1.205 | (0.116) | −0.968 | (0.158) | −1.445 | (0.154) | −1.135 | (0.143) |
| Fraction of negative age coefficients (min. age 61)a | 87.2% | 97.1% | 91.7% | 94.7% | ||||
| I.c. IADL | ||||||||
| Female coefficient | −0.535 | (0.114) | −0.546 | (0.151) | −0.648 | (0.128) | −0.594 | (0.135) |
| Fraction of negative age coefficients (min. age 61)a | 92.3% | 97.1% | 83.3% | 97.4% | ||||
| II. Model includes a dummy for each age and assumes a variable female effect for each age | ||||||||
| II.a. SRHS | ||||||||
| Average value of the female coefficientsb | 0.069 | (0.151) | −0.531 | (0.372) | 0.342 | (0.241) | 0.101 | 0.197 |
| Wald joint test age - female coefficients | 4.13 | p < .0001 | 2.20 | p < .0006 | 79.62 | p < .0125 | 1.44 | p < .0770 |
| II.b. ADL | ||||||||
| Average value of the female coefficientsb | −0.974 | (0.552) | −1.441 | (0.717) | −1.412 | (0.544) | −0.537 | (0.512) |
| Wald joint test age - female coefficients | 47.65 | p < .0000 | 5.44 | p < .0000 | 3.92 | p < .1428 | 18.02 | p < .0001 |
| II.c. IADL | ||||||||
| Average value of the female coefficientsb | −0.331 | (0.389) | −0.782 | (0.279) | −0.306 | (0.545) | −1.066 | (0.345) |
| Wald joint test age - female coefficients | 6.75 | p < .0001 | 4.38 | p < .0001 | 8.98 | p < .0487 | 308.03 | p < .0000 |
| Note. (1) Estimates in bold are significant at the p < .05 level. (2) SRHS was coded 5 = excellent; 4 = very good; 3 = good; 2 = fair; 1= poor. (3) ADL is an indicator from 0 to 10 (0 = worst condition). (4) IADL is an indicator from 0 to 15 (0 = worst condition). OLS = ordinary least squares; SRHS = self-reported health status; ADL = activities of daily living; IADL = instrumental activities of daily living. a Indicates the percentage of age coefficients out of the total with negative sign. b Indicates the average value for the age - female coefficients and standard error. |
||||||||
The regression results from a model specification that includes a dummy variable for each age and assumes a constant female effect (see Equation 1) are very robust and show similar patterns for each country (Table 4). In all countries, after controlling for an individual's age, females show lower self-reported health, ADL and IADL. These results are statistically significant at the .05 level for all measures of an individual's health used in the analysis, with the exception of the finding for the self-reported health indicator in Brazil (ADL and IADL are statistically significant for all countries). Moreover, the fact that the vast majority of coefficients on the age dummy variables have a negative sign indicates that, as expected, health deteriorates with respect to the initial health status reported at 60 years of age. This initial analysis shows that at all ages, elderly females tend to report a lower quality of life (measured by indicators of mobility, ability to perform self-care activities and self-assessment of their own health) than males.
The results from our less-restrictive regression model (see Equation 2) that estimates the effect of gender and age on health status (second part of the results on Table 4) are less robust, but they do suggest that females report lower health than males at each age. First, we report both the average female effect (we calculate the mean value of all the age/female coefficients) as well as its standard error. A t-test was conducted to test the null hypothesis that the average female effect is equal to zero. In almost every instance, we fail to reject this hypothesis. We suspect that this result is due to small sample sizes at each age resulting in female coefficients that are imprecisely measured. Interestingly, in all cases where the average female coefficient value is significant at p < .05, the mean value is negative, indicating that females have lower health on average than males.
Using this specification, we also test the significance of the joint Wald test for the age and gender coefficients. This test could be interpreted as an indication of any of the female effects being different from zero. In the case of self-reported health status, evaluating the joint Wald test is statistically significant at .05 (i.e., suggesting lower health for females for most ages) in the case of Brazil, Argentina and Chile. Yet, for the elderly population in Mexico, this conclusion does not hold. The results using ADL suggest a significant test for the elderly in Brazil, Argentina and Mexico, but not in the case of Chile, while the test using IADL outcomes are insignificant only in Brazil.
Figure 1 summarizes the results of the locally weighted regression analysis for each measure of an individual's health. These findings may be summarized as follows: At almost every age, women report lower self-reported health status than men. Additionally, they also have more problems with ADL and IADL than men, at each age. This is the case for each of the four countries we examined. There is not a single instance in any of the countries, as may be seen in Figure 2, that would lead one to conclude that women, at any elderly age, are in better health than men.
In the case of Brazil, females show lower self-reported health than males at every age; interestingly, the gap in health increases significantly for the elderly above 90 years of age. When one uses ADL as indicator of an individual's health, both males' and females' health status declines with age; however, at all ages females report lower health than males. The gap is virtually constant over all ages. IADL results show a very similar pattern: Males report better outcomes than females at each age, yet, the gap increases with age.
In the case of Argentina, the results for ADL and IADL are similar to the ones in Brazil. Females have lower indicators of mobility and self-care activities than males at all ages. In both cases, the differentials in health are larger for the elderly above 80 years of age than for those younger than 80. When one uses self-reported health, males also show better health than females at all ages. The gender gaps are larger after 80 years of age than they are at younger ages. In Chile, males show better SRHS, ADL and IADL than females at all ages. A woman's health deteriorates with age more rapidly than a man's health for all indicators of an individual's health. Gender differentials in health for the "old old" are bigger than for elderly younger than 85 years of age for all three indicators of health. Lastly, in Mexico, females also show lower health than males at each age. For both males and females, ADL and IADL indicators decline with age. Unlike in the other countries, males' and females' indicator gaps close with age in terms of self-reported health status.
It is important to notice, however, that the lower overall health of females, as shown in Figure 1, could be due to the existence of long-term chronic health conditions that are more likely to exist for females than males. On the other hand, males could be more likely to suffer from more complicated life-threatening conditions, and report higher mortality associated with them despite the fact that they report better ADL and IADL indicators. Furthermore, the existence of better indicators of mobility and self-care could be the consequence of males receiving better support from spouses, and/or other family members. It is plausible that females self-report lower health at each age because they are more likely to visit doctors, and as a result have better knowledge about their health than males. Females could also be less willing to declare themselves in ill-health than males; if so, the results shown in the data could understate the true gender gaps in health. Finally, in addition to gender gaps in health, there could also be gender differences in access to care and the duration and intensity of treatment.
D. What Factors Explain the Patterns of Gender Differentials in Self-Reported Health and Functional Disability among the Elderly?
Biological factors, differences in behavioural choices and differences in reporting symptoms and illnesses have been suggested as explanations for gender differences in health (Verbrugge 1985; Strauss et al. 1993). Using a multivariate analysis, we now turn to the task of evaluating to what extent observable differences explain the gender gaps in health.
For instance, in Section III.B we reported that in all countries included in the study, males are more likely to be married and have lower declines in the rate of marriage than females; males report having higher education levels, are more likely to own their home and report higher incomes than females. All these gender differences could explain why, at all ages, females report lower health than males.
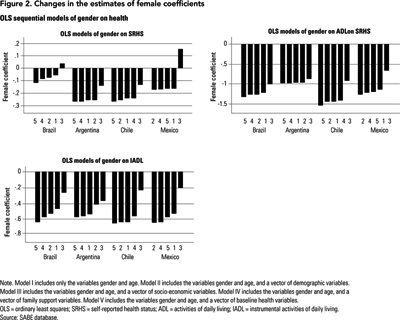
The aim of the ordinary least squares (OLS) sequential analysis described in this section is to identify the individual characteristics that have the greatest impact on reducing these gender gaps in health. The results of this analysis are presented in Table 5 and are illustrated graphically in Figure 2.
| Table 5. OLS sequential regression analysis of the effect of gender and age on the elderly's health; dependent variables – SRHS, ADL, IADL; weighted statistics | ||||||||
| Female coefficient | Brazil | Argentina | Chile | Mexico | ||||
| I. Model includes only gender and age | ||||||||
| I.a. SRHS | −0.058 | (0.040) | −0.259 | (0.068) | −0.245 | (0.078) | −0.159 | (0.052) |
| I.b. ADL | −1.205 | (0.116) | −0.968 | (0.158) | −1.445 | (0.154) | −1.135 | (0.143) |
| I.c. IADL | −0.535 | (0.114) | −0.546 | (0.151) | −0.648 | (0.128) | −0.594 | (0.135) |
| II. Model includes only a vector of demographic variables | ||||||||
| II.a. SRHS | −0.073 | (0.044) | −0.256 | (0.068) | −0.263 | (0.072) | −0.173 | (0.061) |
| II.b. ADL | −1.249 | (0.121) | −0.891 | (0.167) | −1.398 | (0.163) | −1.251 | (0.154) |
| II.c. IADL | −0.485 | (0.108) | −0.368 | (0.155) | −0.575 | (0.131) | −0.651 | (0.145) |
| III. Model includes only a vector of socio-economic variables | ||||||||
| III.a. SRHS | 0.038 | (0.051) | −0.145 | (0.073) | −0.132 | (0.096) | 0.156 | (0.092) |
| III.b. ADL | −0.9967 | (0.138) | −0.965 | (0.178) | −0.924 | (0.225) | −0.669 | (0.268) |
| III.c. IADL | −0.265 | (0.112) | −0.416 | (0.181) | −0.237 | 0.248 | −0.205 | (0.275) |
| IV. Model includes only a vector of family support variables | ||||||||
| IV.a. SRHS | −0.083 | (0.040) | −0.265 | (0.066) | −0.243 | (0.090) | −0.166 | (0.052) |
| IV.b. ADL | −1.249 | (0.117) | −0.969 | (0.156) | −1.432 | (0.160) | −1.189 | (0.139) |
| IV.c. IADL | −0.581 | (0.113) | −0.571 | (0.148) | −0.662 | (0.129) | −0.639 | (0.133) |
| V. Model includes only a vector of baseline health variables | ||||||||
| V.a. SRHS | −0.115 | (0.041) | −0.275 | (0.070) | −0.269 | (0.078) | −0.161 | (0.056) |
| V.b. ADL | −1.317 | (0.121) | −0.976 | (0.154) | −1.529 | (0.157) | −1.217 | (0.144) |
| V.c. IADL | −0.651 | (0.114) | −0.581 | (0.147) | −0.649 | (0.137) | −0.534 | (0.139) |
| VI. Model includes all variables | ||||||||
| VI.a. SRHS | −0.018 | (0.057) | −0.171 | (0.076) | −0.125 | (0.095) | 0.063 | (0.105) |
| VI.b. ADL | −1.099 | (0.141) | −0.951 | (0.185) | −0.897 | (0.218) | −0.635 | (0.262) |
| VI.c. IADL | −0.292 | (0.139) | −0.224 | (0.182) | −0.254 | (0.251) | −0.012 | (0.322) |
| Note. (1) Estimates in bold are significant at the p < .05 level. (2) Standard errors in parenthesis. (3) The vector of demographic variables includes total children alive, currently married and number of marriages. (4) The vector of socio-economic variables includes literacy, education, years of education, home ownership, wealth and total income. (5) The vector of family support variables includes number of household member and number of siblings. (6) The vector of baseline health variables includes living in rural areas <5 years, smoking, initial family economic condition, initial self-assessment of health and initial starvation. (7) All models included a separate dummy for each age and assumed a constant female effect. OLS = ordinary least squares; SRHS = self-reported health status; ADL = activities of daily living; IADL = instrumental activities of daily living. |
||||||||
As shown in Table 5, for the basic model the results are consistent with a gender gap in health in favour of males, after controlling for an individual's age. For all measures of health, females report lower health than males, and these results are significant at the .05 level with the only exception being the model that uses the SRHS in the case of Brazil.
These gender differentials in health, observed in the basic models, could be the result of elderly females being less likely to be married than elderly males (as our previous analysis suggests), or because they have fewer children alive who could provide protective support for them. In Model II, however, we learn that controlling for these differences in demographic characteristics does little to reduce these gender gaps in health. For most cases, the female coefficients are still very close to that found in the basic model; all are still negative and most are significant at the .05 level. A similar result occurs when we include the vector of family support variables (Model IV) and baseline health conditions (Model V). Essentially, comparing seniors with similar baseline conditions and family support characteristics does not eliminate the original gender differences between males and females. Although these are factors that influence an individual's health, and therefore could be the driving force behind the gender gaps in health as observed in the data, the findings suggest that eliminating demographic and baseline health differentials in health or equalizing family support conditions will not do much to reduce the health differentials between males and females.
While controlling for demographic characteristics, baseline health or the availability of family support does little to change the health disadvantage for women in measured health outcomes, controlling for socio-demographic variables (Table 5, Model III) in almost every instance reduces the gender difference for each of the three health outcome measures. Figure 2 graphs the estimated female coefficient for each model. For all measures of health, Model III, which includes socio-economic variables, yields the smallest gender effect on health. Results for Argentina, when one uses ADL or IADL, are the only exceptions.
Interestingly, in developed countries, gender gaps in socio-economic conditions among the elderly have been reported as having little relevance in explaining differences in health among elderly males and females (for example, in the case of the United States, see Strauss et al. 1993). Two alternative explanations have been suggested for this observation. First, the lack of relevance of socio-economic conditions to explain an individual's health is the consequence of a selection process. Second, the institutional design of the healthcare system may reduce the influence of socio-economic variables on an individual's health.
The selection argument implies that socio-economic differences matter less and less as we compare the health of older individuals, if sicker individuals, who die earlier, are also the ones with lower income, wealth and education. Second, the existence of a medical care system that provides access to care regardless an individual's economic status will make differences in socio-economic conditions less relevant in explaining gender gaps in health among the elderly. For example, in the case of the United States, the Medicare health insurance system is an entitlement public program that provides access to medical care to all individuals above 65 years of age. Clearly, within the context of this institutional setting, it is reasonable to expect that differentials in socio-economic conditions will exert less of an influence on the gender gaps in health than would be the case in an open-ended insurance scheme.
We presume that issues related to differentials in access to care and quality of medical care with respect to an individual's income may partially explain our findings. For instance, since males have a better financial situation, they may be more likely to use private healthcare providers than females. Further research is warranted, however, to gain a better understanding of the causal links between socio-economic conditions and the observed gender gaps in health.
IV. Concluding Remarks and Policy Implications
This study provides a detailed description of the health conditions by gender among the elderly population in four Latin American cities. Policies designed to increase the elderly females' overall quality of life and ability to function in these economies could benefit by using this information. Yet, one problem with the SABE data might be that it considers only individuals living in select urban areas, ignoring those living in rural areas. The generalizability of these findings should be tempered if considering the potential that the analysis has for informing policy.
The results of this study show that males report better health status than females in all four countries, and females suffer more frequently from disabling conditions, physical functioning, hypertension, diabetes and cancer. Females also report lower cognitive score than males. A cross-country comparison indicates that females in Argentina show the highest prevalence of disabling conditions. Chileans report the lowest rate of diabetes but the highest prevalence of heart diseases. Lastly, Mexicans report the lowest rate of hypertension and show a high prevalence of diabetes.
Our findings regarding the gender gaps in health, however, must be interpreted with caution. In particular, the lower prevalence of life-threatening conditions among males could be the result of males having a lower tendency to visit the physician and/or a decreased awareness of their health status. One possible cost-effective intervention would involve targeting low-income males to improve their access to medical care.
The gender gaps in self-reported and functional health in this analysis could be biased for several reasons. First, it has been reported that in many developing countries females are less likely than males to report themselves in ill-health (see, for example, Saith and Harriss-White 1999). If this is the case, the gender gaps reported in our analysis underestimate the true differences in morbidity between the male and female elderly in these cities. Second, the data about prevalence of health conditions might reflect information about those who have received medical care. Thus, these data do not reflect the true incidence of diseases, but rather the availability of medical treatment. Lastly, gender differences in the duration and intensity of the medical condition, and the potential gaps in received treatment, are not reported in the data.
Our empirical analyses suggest that gender differentials in age composition are not the main reason behind these results. In particular, at almost every age, women have lower self-reported health status than men. In addition, they also have more problems with ADL and IADL than men at each age.
Controlling for demographic characteristics, baseline health or the availability of family support does little to change the disadvantage faced by women in measured health outcomes at each age. Controlling for socio-economic variables in almost every instance reduces the gender differences in health. The strength of the results suggests that socio-economic differences in literacy, education, income and wealth may be translated into affecting individual behaviours that improve seniors' health. Yet, using these data we could not identify the precise channels by which socio-economic differences translate into better health. One possible channel could be the differences in access to care and quality of care by socio-economic status. We could not identify how biological differences in reporting symptoms and following medical treatment play a role in explaining the gender gaps in health observed in the data. Future research could be directed in this area in an effort to gain a better understanding of these factors and their relationships with seniors' health in developing countries.
Integrating all of the aforementioned results suggests that health policies aimed at increasing the well-being (i.e., health status) of females and their access to medical care should be combined with income policies that reduce gender differentials in socio-economic status among seniors. Another policy implication of these results is that analysts should focus on policies that increase quality of life rather than just reduce gender gaps in mortality. The fact that our findings are very similar across different Latin American countries with different healthcare systems, economic situations and cultural backgrounds suggests that the gender gaps in health are the consequence of structural common gender differences across countries.
Analysts could develop primary prevention programs to reduce the prevalence of disabling conditions throughout the population, with special targeting toward females. Chronic disease management practices would also reduce prevalence and medical care costs associated with life-threatening conditions, such as cancer, diabetes, hypertension and others. Buvinic et al. (2006) propose a list of cost-effective interventions targeted at improving females' health. These practices could free up public resources to improve the efficiency with which medical care and services are delivered. The Chilean experience with diabetes and the Mexican experience with hypertension could provide an initial framework for policy development in other countries. Additional policies that would improve the health status of seniors in these countries might include promoting healthy lifestyles, improving early-life conditions and nurturing interpersonal relationships within the family unit and within the community.
About the Author(s)
Antonio J. Trujillo, PhD, Department of International Health, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland
Thomas A. Mroz, PhD, John E. Walker Economics Department, Clemson University, Clemson, South Carolina
Claudia Piras, Inter-American Development Bank, Washington, DC
John A. Vernon, PhD, Department of Finance, University of Connecticut, Storrs, Connecticut
Gustavo Angeles, School of Public Health, Department of Maternal and Child Health, University of North Carolina, Chapel Hill, North Carolina, Instituto Nacional de Salud Pública, México
Antonio J. Trujillo, MPP, PhD, Department of International Health, Johns Hopkins Bloomberg School of Public Health, 615 North Wolfe Street, Suite E-8132, Baltimore, MD 21205, USA. Phone: +1- 443-287-3003, Email: atrujill@jhsph.edu
Acknowledgment
We thank the Inter-American Development Bank, Sustainable Development Department, Gender Equality in Development Unit for the financial support provided for this investigation. We also thank Jennifer Kasino for excellent research assistance during the development of this project. The authors take sole responsibility for any remaining errors. We would like to acknowledge the principal investigators who participated in the SABE project and the Pan-American Health Organization for providing the data used in this analysis.
References
Arber, S. and H. Cooper. 1999. "Gender Differences in Health in Later Life: the New Paradox?" Social Science and Medicine 48: 61–76.
Arriagada, I. 2003 Transformaciones Sociales y demográficas de las familias latinoamericanas. Working Paper Inter-American Development Bank. 1–20.
Bos, A. and A. Bos. 2005. The Social–Economic Determinants of Elderly Health in Brazil: the Importance of Marital Status and Income. World Bank.
Buvinic, M., A. Medici, E. Fernande and A.C. Torres. 2006. Gender Differentials in Health. Social Inclusion and Economic Development in Latin America, 1st Edition pp. 195-210. Inter-American Development Bank.
Deeg, D.J.H. and D.M.W. Kriegsman. 2003. "Concepts of Self-rated Health: Specifying the Gender Difference in Mortality Risk." The Gerontologist 43(3): 376–86.
Grossman, M. 1972. "On the Concept of Health Capital and the Demand for Health." Journal of Political Economy 80: 223–55.
Hurd, M.D., D. MacFadden and A. Merrill. 1999. Predictors of Mortality among the Elderly. NBER Working Papers 7440, National Bureau of Economic Research, Inc.
Iacovou, M. 2000. The Living Arrangements of Elderly Europeans. ISER Working Papers, Institute for Social and Economic Research.
Knodel, J. and M.B. Ofstedal. 2003. "Gender and Aging in the Developing World: Where Are the Men?" Population and Development Review 29(4): 677–98.
Nathanson, C.A. 1975. "Illness and the Feminine Role: a Theoretical Review." Social Sciences and Medicine 9(2): 57–62.
Ross, C.E. and C.E. Bird. 1994. "Sex Stratification and Health Lifestyle: Consequences for Men's and Women's Perceived Health." Journal of Health and Social Behavior 35(2): 161–78.
Saad, P.M. 2002. Transferencias Informales de Apoyo de los Adultos Mayores en América Latina y el Caribe: Estudio Comparativo de Encuestas SABE. Unpublished manuscript. . 175–218.
Saith, R. and B. Harriss-White. 1999. "The Gender Sensitivity of Well-being Indicators." Development and Change 30(3): 465–97.
Sen, A. 1985. Commodities and Capabilities. Amsterdam: Elsevier Science Publishers.
Sen, A. 1998. "Mortality as an Indicator of Economic Success and Failure." The Economic Journal 108(446): 1–25.
Shrestha, L.B. 2000. "Population Aging in Developing Countries." Heath Affairs 19(3): 204–12.
StataCorp. 2003. Stata Statistical Software: Release 8. College Station, TX: StataCorp.
Strauss, J., P.J. Getler, O. Rahman and K. Fox. 1993. "Gender and Life-Cycle Differentials in the Patterns and Determinants of Adult Health." Journal of Human Resources 28(4): 791–837.
Valanis, B. 1999. Epidemiology in Health Care. 3rd Edition. Stamford, Connecticut: Appleton & Lange.
Verbrugge, L.M. 1985. "Gender and Health: an Update on Hypotheses and Evidence." Journal of Health and Social Behavior 26(3): 156–82.
Footnotes
1. Other indicators of well-being would be education and nutrition outcomes. For more details about this approach, the reader should consult Saith and Harriss-White (1999) and Sen (1998, 1985).
2. For a detail description of differences in health among males and females during childhood and adult life, the reader should review the articles by Verbrugge (1985), Strauss et al. (1993), Ross and Bird (1999) and Valanis (1999).
3. Using longitudinal data, one could explore this issue by evaluating the health of those males and females who die during the period of data collection.
4. For more detail about this procedure, the reader should review the Stata manual, Version 8 (Stata Corp 2003).
5. In the next section, we describe which variables are included in each vector.
6. The SABE web page (www.ssc.wisc.edu/sabe) lists complete information on the agencies and researchers who participated in this project.
7. Table 1 is a relevant, but partial, list of variables available from the long version of the SABE survey.
Comments
Sophie Chiasson wrote:
Posted 2010/11/29 at 10:25 PM EST
good
Personal Subscriber? Sign In
Note: Please enter a display name. Your email address will not be publically displayed