Healthcare Policy
Public Drug Plan Coverage for Children Across Canada: A Portrait of Too Many Colours
Abstract
Background: As debate continues regarding pharmacare in Canada, little discussion has addressed appropriate drug plan coverage for vulnerable populations, such as children. The primary objective of this study was to determine the extent of medication coverage for children in publicly administered programs in each province across Canada.
Methods: Data were collected on provincial, territorial and federal government drug plans, and 2003 formulary updates were obtained. A simulation model was constructed to demonstrate costs to a low-income family with an asthmatic child in each province. Programs were compared descriptively. The extent of interprovincial variation in 2003 formulary approvals was summarized statistically.
Results: There was 39% variation between provinces with respect to 2003 formulary approvals (chi-square p < 0.0001) and 48% variation for 2003 paediatric-labelled products (chi-square p < 0.0001). Across Canada, only 8% of 2003 formulary approvals were indicated primarily for paediatric conditions. In the simulation model, costs were less than or equal to 3% of household income in provinces with plans for low-income families, catastrophic costs (Ontario) or for the population. Families who failed to qualify for low income plans or who resided in New Brunswick or Newfoundland faced costs up to 7% of household income.
Interpretation: With regard to pharmaceutical benefits for children, provincial drug programs vary considerably in terms of whom they cover, what drugs are covered and how much subscribers must pay out of pocket. Unlike seniors and social assistance recipients, the provinces do not agree on the importance of providing comprehensive coverage for all children. For many Canadian children, significant financial barriers exist to medication access.
[To view the French abstract, please scroll down.]
A spate of reports scrutinizing the healthcare system in recent years (Health Canada 1997; Kirby 2002; Romanow 2002) have brought new life to the discussion concerning a national pharmacare program. A program that would ensure access to and affordability of needed medications, particularly for vulnerable populations, has repeatedly been cited as a priority by policy makers as well as stakeholder groups. This goal has yet to be realized, and the debate regarding what constitutes optimal pharmaceutical policy in Canada continues. Currently in Canada, payment for prescription medicines is financed by a combination of public and private sources. In 2001, public plans, consisting of provincial and territorial drug programs, accounted for 46% of total prescription drug spending in Canada (Canadian Institute for Health Information 2004). Persons aged 65 and older accounted for 65% of the $4.44 billion spent on public drug programs in 2001. In contrast, persons aged 14 and under accounted for 2.1% (Health Canada 2001). While provincial policy makers agree on the importance of providing medication benefits to seniors, there is no agreement on the need to provide the same benefits to other vulnerable populations, including Canada's 7.5 million children. Thus, the low public spending on pharmaceutical benefits for children may reflect a lack of programs to meet children's needs.
A number of reports have exposed differences in provincial drug plan characteristics related to eligibility, cost-sharing and listed benefits (Canadian Institute for Health Information 2004; Health Canada 2000; Jacobs and Bachynsky 2000; Grootendorst 2002; Narine and Sen 1997; Currie and Nielson 1999; Willison et al. 1998; Morgan 2004). However, none has focused on access to benefits for the paediatric population. The primary objective of this study was to determine the extent of medication coverage for children in publicly administered programs in each province across Canada. This study also investigated the proportion of new drugs added to each provincial formulary in 2003 that included indications for paediatric conditions or allowed prescribing for children.
Methods
Data sources
All data were collected from primary government sources from January to April 2004. Initially, individual provincial, territorial and federal government websites were evaluated for information and details regarding public drug plans. The information collected included program names and types, eligibility requirements, amounts of premiums, deductibles and co-payments, details of plan restrictions and separate formularies. If the information required was not available from a government website, an email request or phone inquiry was made or a letter of request was sent. Appropriate provincial ministry representatives were identified through Web contacts and telephone calls. All information found on the websites was double-checked as often as required, using phone interviews with representatives of the provincial ministries of health. Where necessary, managers of specific or special programs were also contacted to inquire about and validate information. Useful secondary sources of information on provincial drug programs include the report, Drug Expenditures in Canada (Canadian Institute for Health Information 2004), Provincial Drug Benefit Programs (Canadian Pharmacists Association 2004) and the 2004 Guidebook on Government Prescription Drug Reimbursement Plans and Related Programs (Canadian Association for Pharmacy Distribution Management 2004).
Asthma simulation model
The various public drug plan characteristics were illustrated in a scenario analysis that simulated the out-of-pocket expenditures incurred in each province by a low-income, two-parent family with two children, in which one child suffered from moderate to severe asthma. The scenario was simulated for two or three levels of low annual household income for each province, typically $20,000 and $24,000, as these thresholds best exemplified expenditures when families met or failed to meet eligibility for benefits. A typical one-year treatment regimen in compliance with Canadian guidelines (Ernst et al. 1996) assuming optimal adherence was constructed and included:
- Flovent Diskus™ (fluticasone), 250 micrograms per inhalation, 1 puff BID, 60 blister pack, annual requirement of 12 packs
- Ventodisk™ (salbutamol), 200 micrograms per inhalation, administered as needed, 8 blister pack, 15 packs per carton, annual requirement of 2 cartons
- Serevent Diskus™ (salmeterol), 50 micrograms per inhalation, 1 puff BID, 60 doses per inhaler, annual requirement of 12 inhalers
For ease of comparison, a constant medication-regimen price was assigned based on the average of 2004 listed formulary prices for Ontario, Quebec, Saskatchewan and Alberta. The total price was inflated by a 10% allowable markup and a dispensing fee of $6.54 per refill was added, except for Prince Edward Island, where the provincial dispensing fee constituted the fixed co-pay in low-income families. The scenario analysis was based on 2004 total household income, and the following assumptions were made:
- The family's income was too high to qualify them for social assistance
- The family had no private insurance
- The children were not wards of the state
- The child's asthma drugs were the family's only prescription medications
- The family was aware of and made full use of provincial benefit plans where eligible
- The application process for participation in benefit plans was not a deterrent
Flovent Diskus™ and Ventodisk™ were listed as benefits in all provinces. Serevent Diskus™ was listed as a benefit in New Brunswick, Quebec, Manitoba, Alberta and the Yukon and as limited use in Ontario, Prince Edward Island, Nova Scotia, Saskatchewan and British Columbia. It was assumed that when a family qualified for benefits, the plan would pay for Serevent Diskus™ in those provinces where it was designated as limited use. This drug was not approved in Newfoundland.
Comparison of 2003 formulary updates
Formulary updates for 2003 were obtained for each province and territory, except Nunavut and the Northwest Territories. The 2003 formulary for the federal government plan, which covers First Nations residents across Canada and the armed forces, was also obtained. Where possible, a list of drugs added to a provincial formulary in 2003 was acquired directly from a representative of the respective ministry of health. Otherwise, under the advisement of ministry of health representatives, the formulary updates/bulletins for 2003 were used to create a database of all the drugs added to the provincial formulary in 2003. The individual 2003 formulary updates were compiled in a single database of all the prescription and non-prescription drugs added to the provincial and territorial formularies across Canada in 2003. It is possible that drugs that were added in 2003 in a given province may have already been listed in other provinces. No adjustments to the database were made for these drugs. Prescription diabetic medications and enteral nutritional products were included, whereas diabetic and injection supplies, such as test strips, glucometers, needles, lancets and syringes were excluded. Fibre supplements, electrolyte solutions, dermatological products, anti-venom agents, masks and devices were excluded.
Listed drugs were flagged if they were approved for use in children or if their primary use was for a child's condition. The term "paediatric-labelled" is used to indicate medications that fulfill either of these two criteria. The 2003 Compendium of Pharmaceuticals and Specialties (Canadian Pharmacists Association 2003), Mosby's Drug Guide for Nurses (2003), drug monographs and information provided by drug manufacturers, as well as several Internet databases, including the Drug Product Database - Health Canada, and Medline Plus - the National Library of Medicine, were used to verify drug identification numbers, drug names, drug formulations and details of labelling.
Statistical analysis
Analyses were conducted on the full dataset of formulary additions, including multiple dosage forms and strengths, and not just on new chemical entities. This approach was chosen because the variety of formulations and strengths available for any particular drug relates to the extent of access to that medication. Descriptive statistics were used to describe differences in volume of listed products, products approved for use in children and products with a mainly paediatric indication across provinces and territories. The coefficient of variation, which is the standard deviation divided by the mean, and the extremal quotient (EQ), the ratio of maximum to minimum, were computed as point estimates of interregional variation for each variable. A correction factor of 0.02 was used in the case of zero-value denominators. The statistical significance of variation for each variable was measured with a chi-square test, which compared observed variation to the mean across all provinces.
Results
Provincial drug program characteristics
The provincial prescription drug programs that provide medication benefits to children across Canada vary considerably. Table 1 summarizes the types of programs in which children are included or are the main focus. For many plans, benefits extend to whole families, and children gain access through their parents who meet the eligibility criteria. This applies to social assistance programs, income-indexed drug plans and special plans for low-income families. In addition, some provinces offer special programs for children with chronic diseases such as cystic fibrosis or who are severely handicapped. The prescription drugs paid for through the various provincial programs are those listed in the respective provincial formularies.
| Table 1. Drug Programs that provide benefits to children | ||||||||||||
| NL | PE | NS | NB | QC | ON | MB | SK | AB | BC | YT | NT | NU |
|
Universal program for all residents without private insurance. Deductibles not income-indexed. |
||||||||||||
| √ | √ | |||||||||||
| Income-indexed drug plan | ||||||||||||
| √ | √ | |||||||||||
| Income-indexed catastrophic drug plan for persons with very high costs relative to income or transitional plan for persons leaving social assistance | ||||||||||||
| √ | √ | √ | √ | √* | ||||||||
| Social assistance/Welfare | ||||||||||||
| √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | √ | ||
| Special family/child program for low-income families | ||||||||||||
| √ | √ | √ | √ | |||||||||
| Specific program for: | ||||||||||||
| - Cystic fibrosis | ||||||||||||
| √ | √ | √ | √ | |||||||||
| - Diabetes | ||||||||||||
| √ | √ | |||||||||||
| - Human Growth Hormone | ||||||||||||
| √ | √ | √ | ||||||||||
| - Severely handicapped children | ||||||||||||
| √ | √ | √ | ||||||||||
| - Umbrella program for chronic disease like CF | ||||||||||||
| √ | √ | √ | √ | √ | √ | |||||||
Programs described are those in effect as of April 2004. *Alberta's transitional program is not income-indexed and does not require cost-sharing. |
||||||||||||
Only Quebec and Alberta provide universal coverage, defined as coverage for all residents of the province who are not privately insured. While cost-sharing exists in these plans, deductibles are not income-indexed. In Quebec, all forms of cost sharing are waived for individuals aged less than 18 years. Six provinces offer plans with cost-sharing arrangements that require subscribers to pay deductibles or co-payments that are tied to their household income or both. The catastrophic and transitional drug plans are a sub-set of the income-indexed plans, except for Alberta, which has a transitional program without cost-sharing. The line between regular income-indexed plans and "catastrophic" plans is thin and is essentially a function of the amount of cost-sharing required. The Manitoba plan, for example, is for persons "whose income is seriously affected by high prescription drug costs" (Manitoba Health 2004). "Catastrophic" plans are typically characterized by a requirement that drug costs be a substantial portion of income and by their very large deductible - a perverse arrangement, given that these plans are designed for those with the greatest medication needs. Transitional programs are plans that provide benefits to individuals leaving social assistance to return to the workforce. High deductibles and co-payments in catastrophic and transitional plans can pose a significant financial barrier, particularly for families with several members requiring multiple prescription medications, as in families where several children are afflicted with asthma. A description of cost-sharing components of specific plans, including premiums, deductibles, co-payments and restrictions, can be found in Table 2.

All provinces have a program for poor families receiving government assistance. None of these programs has a maximum annual benefit, but each province has different eligibility criteria, as the definition of low income varies. Also, some provinces waive the deductibles and co-payments for children's medications, as is the case for Saskatchewan families receiving social assistance and residents of Quebec.
Prince Edward Island, Saskatchewan, Alberta and Yukon have programs that are exclusively meant for children of low-income families who are not under the care or custody of the government. In Prince Edward Island and Saskatchewan, the programs offer access to the provincial formulary for children of families whose incomes qualify them. In Alberta, the Child Health Benefit program provides similar benefits for children of low-income families. There are no fees or annual maximum benefit restrictions associated with the program, and if the parents have private insurance this program will pay the co-payment. In Yukon, the Children's Drug and Optical Program provides access to prescription medications to children from low-income families. There are no premiums, co-payments or maximum annual benefit restrictions with this plan. Further, there are no deductibles for very low-income families or low-income large families. However, other families face annual deductibles, which reach a maximum of $500 per family.
Some provinces and territories, namely Ontario, Saskatchewan, Alberta, Yukon, the Northwest Territories and Nunavut, have an "umbrella" special program for various chronic diseases occurring in adults and children. These special programs usually have a separate drug benefit list from the provincial formulary, but if the indicated drug is found on the provincial formulary, it may be denoted as available only to clients of the special program. To be eligible, the client has to be clinically diagnosed with the condition that the program covers, without having to fulfill any financial or other criteria. Only the Yukon program has a fee associated with it, a $250 deductible, for a maximum of $500 per family.
In Ontario, the Special Drugs Program pays for drugs to treat cystic fibrosis, thalassaemia and growth failure due to insufficient growth hormone. In Saskatchewan, this type of program is called SAIL (Saskatchewan Aids to Independent Living) and covers only one paediatric condition, namely, cystic fibrosis. In Alberta, the Province Wide Services program covers drugs for cystic fibrosis and paediatric growth hormone deficiency. In Yukon, the Chronic Disease Program provides drug coverage for several paediatric conditions such as attention deficit/hyperactivity disorder, cystic fibrosis, diabetes and others. In the Northwest Territories, the Extended Health Benefits for Specified Diseases program covers many paediatric conditions, including asthma, cystic fibrosis and spina bifida, among others.
In contrast to the chronic disease programs described above that serve both adults and children, Newfoundland, Prince Edward Island, New Brunswick, Saskatchewan and British Columbia have set up special programs for specific paediatric conditions. In Newfoundland, the Special Needs Program provides coverage for cystic fibrosis patients, and persons requiring growth hormone or special foods because of a metabolic disorder such as phenylketonuria. The program provides prescription drugs and any other necessary supplies. Prince Edward Island has individual programs for children suffering from diabetes, cystic fibrosis, growth hormone deficiency and meningitis. Prince Edward Island also provides a Nutrition Services Program for children at risk for nutritional deficiency and a Phenylketonuria Program. The Prince Edward Island programs have no cost or annual maximum benefits associated with them. New Brunswick has special programs for cystic fibrosis patients (Plan B) and for individuals with growth hormone deficiency (Plan T). Both programs have a yearly fee of $50 and a co-payment of 20% or a maximum of $20 (annual maximum of $500 per family). British Columbia has a specific program for cystic fibrosis patients (Plan D) that provides medications at no cost.
In addition to the above programs, New Brunswick, Ontario, British Columbia and Yukon have established programs for children with severe disabilities. Typically, these programs evaluate children's health and other needs on a case-by-case basis.

Asthma simulation model
The scenario analysis in Table 3 indicates the out-of-pocket expenditures for low-income households in each province where one child requires multiple prescription medications for treatment of moderate to severe asthma. This scenario was selected as asthma is a common chronic health problem in children for which multiple expensive medications are routinely prescribed (Millar 1998; Mannino et al. 1998; Kozyrskyj et al. 2001). The scenario was simulated for two or three levels of low annual household income for each province, typically $20,000 and $24,000, as these thresholds often provided an informative contrast when families met or failed to meet eligibility for benefits. In many provinces or territories where plans existed for low-income families (Saskatchewan, Yukon), for catastrophic drug costs (Ontario) or for the population at large (Quebec, Manitoba, Alberta, British Columbia), total out-of-pocket expenditures remained less than or equal to 3% of household income. In these provinces, low-income families received reasonably good coverage, regardless of their income level, assuming they knew about the program and were successful in applying. In Prince Edward Island and in Nova Scotia, families who met the eligibility for low income (less than $24,000) or who were within one year of receiving social assistance benefits, respectively, faced low financial barriers. Those low-income families who failed to meet eligibility in these provinces, or those who resided in New Brunswick or Newfoundland, faced formidable financial barriers, with out-of-pocket expenditures reaching up to 7% of household income.
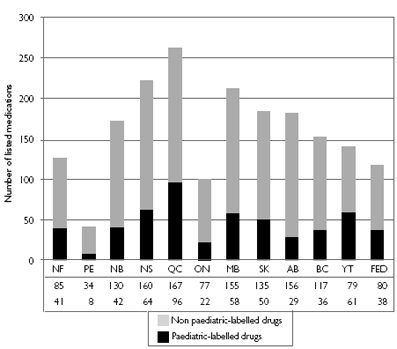
| Figure 1. Medications approved for formulary listing in 2003
Paediatric-labelled drugs include all medications approved for use in children in 2003. Variation in number of drugs listed: coefficient of variation = 39.3%, extremal quotient = 6.3, chi-square p < 0.0001. Variation in number of paediatric-labelled drugs: coefficient of variation = 48.4%, extremal quotient = 12.1, chi-square p < 0.0001. |
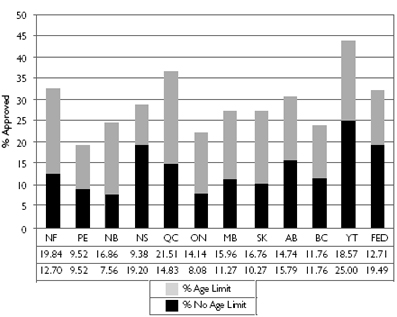
| Figure 2. Proportions of listed drugs approved for use in children
% Age Limit indicates the proportion of drugs listed in 2003 that were approved for use in children with a minimum age requirement. % No Age Limit refers to the proportion of drugs listed in 2003 that were approved for use in children with no minimum age requirement. Variation in % of total paediatric-labelled drugs: coefficient of variation = 22.3%, extremal quotient = 2.3. Variation in % of listed drugs with age limit: coefficient of variation = 24.4%, extremal quotient = 2.3. Variation in % of listed drugs with no age limit: coefficient of variation = 36.6%, extremal quotient = 3.3, chi-square p < 0.05. |
Access to paediatric-labelled products
In 2003, 754 products were added on a cumulative basis to provincial/territorial formularies across Canada. The majority of these were multi-sourced, interchangeable, generic products with identical active ingredients in multiple strengths and formulations or were incrementally modified drugs (IMDs) consisting of the addition of a new strength or dosage form to an existing product. Only 265 additions (35%) were unique chemical entities, most of which were IMDs. Of all the drugs cumulatively approved in 2003 for listing across Canada, 271 (36%) were approved for use in children. Of these 271, 54% were labelled with a minimum age requirement. Among the unique chemical entities, 122 (46%) were approved for use in children. Of these 122, 50% were labelled with a minimum age requirement. As seen in Figure 1, there was 39% variation between the provinces/territories with respect to the number of new products listed in 2003 (chi-square p < 0.0001). The EQ indicates that there was a sixfold difference between the minimum and maximum, 42 in Prince Edward Island versus 263 in Quebec. There was even greater variation (48%) with respect to the number of new paediatric-labelled products listed in 2003 (chi-square p < 0.0001) with an EQ indicating a 12-fold difference between the minimum of eight drugs in Prince Edward Island and 96 in Quebec. As seen in Figure 2, of the products listed to individual formularies, Yukon, Quebec and Newfoundland had the greatest proportions of drugs approved for use in children, with 44%, 36% and 33%, respectively. Prince Edward Island, Ontario and British Columbia had the lowest proportions of newly listed drugs approved for use in children, with 19%, 22% and 24%, respectively. The proportions of listed products with age restrictions varied from 9% in Nova Scotia to 22% in Quebec.
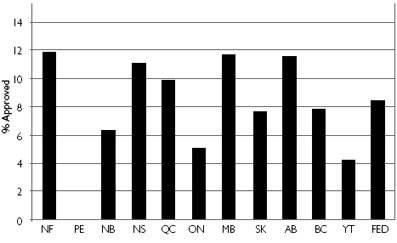
| Figure 3. Proportions of listed drugs approved for mainly paediatric conditions
Variation in % of drugs approved in 2003 for paediatric conditions: coefficient of variation = 44%, extremal quotient (corrected) = 235. |
Of the cumulative number of drugs approved for Canadian public formularies in 2003, only 8% (61/754) were indicated primarily for paediatric conditions. Figure 3 illustrates that there was 43% variation across the provinces, ranging from a high of 12% in Newfoundland, Manitoba and Alberta to lows of 0% in Prince Edward Island, 4% in Yukon and 5% in Ontario.
| Table 4. Costs and listing status for select paediatric medications | ||||||||||||
| Weekly Cost | NL | PE | NS | NB | QC | ON | MB | SK | AB | BC | YT | Fed. |
| Drug: Desferal™ (deferoxamine mesylate) Indication: chronic iron overload |
||||||||||||
| $183.75 | NC | NC | GB | GB | GB | NC | GB | LU | GB | GB | NC | NC |
| Drug: Enbrel™ (etanercept) Indication: juvenile rheumatoid arthritis |
||||||||||||
| $375.00 | NC | NC | LU | LU | GB | NC | NC | LU | LU | NC | LU | LU |
| Drug: Neupogen™ (filgrastim, GCSF) Indication: febrile neutropenia in patients with nonmyeloid malignancies |
||||||||||||
| $964.18 | NC | NC | NC | LU | LU | NC | GB | LU | LU | NC | LU | GB |
| Drug: CellCept™ (mycophenolate mofetil) Indication: prophylaxis of organ rejection in children receiving allogeneic renal transplants |
||||||||||||
| $230.95 | NC | GB | NC | NC | GB | LU | LU | LU | NC | NC | GB | GB |
| Drug: Zofran™ (ondansetron) Indication: prevention of nausea and vomiting associated with chemo and radiotherapy |
||||||||||||
| $125.76 | NC | LU | LU | LU | LU | LU | GB | NC | GB | NC | LU | GB |
| Drug: Tazocin™ (piperacillin sodium & tazobactam sodium) Indication: antibacterial |
||||||||||||
| $333.90 | NC | NC | NC | NC | GB | NC | NC | NC | GB | NC | NC | NC |
Abbreviations: GB = General Benefit; LU = Limited Use; NC = Not Covered Listing status and unit prices are as of September 2004. Costs are based on recommended maintenance dosage regimens for a 40 kg child |
||||||||||||
The variation in listing status is exemplified by decisions regarding expensive, but efficacious, medications. Table 4 lists six medications for which outpatient access was deemed medically necessary by clinical experts at the Hospital for Sick Children. While some of the more expensive medications, such as Neupogen™ and Tazocin™, are typically administered for short durations, administration of drugs such as Enbrel™ and CellCept™ may continue for months or longer, causing economic hardship to families without access to adequate pharmaceutical benefits. As of September 2004, Enbrel™, a treatment for juvenile rheumatoid arthritis, was a general benefit in one province/territory, a limited-use benefit requiring prior authorization by a physician in six provinces/territories and was not covered in five provinces/territories. This drug costs $19,500 for one year of treatment. Neupogen™ is used to prevent neutropoenia in children receiving myelosuppressive chemotherapy. This drug, which costs $11,500 for four courses of treatment, is available as a general benefit in two provinces/territories, as a limited-use product in five provinces/territories and is not covered in five provinces/territories. Children with end-stage renal disease who require Desferal™ for treatment of iron overload are fortunate if they live in one of the six provinces/territories where this product is a general benefit. This product is available as limited use in one province/territory but is not covered in five other provinces/territories. While these medications can sometimes be obtained by special authorization in provinces that do not provide coverage, the application process for such authorization can be lengthy and onerous, and there is no guarantee that the request will be approved. The listing pattern for these drugs was similar to that seen for all medications in Figure 1, with Quebec, Manitoba and Alberta demonstrating the greatest access and Ontario, British Columbia, Newfoundland and Prince Edward Island showing the poorest access.
Interpretation
These findings indicate that with regard to providing pharmaceutical benefits to children, provincial drug programs vary considerably in terms of whom they cover, what drugs are covered and how much subscribers must pay out of pocket. In addition, the majority of drugs listed on provincial formularies are not labelled for use in children, and even fewer are indicated for paediatric conditions.
Variation in plan eligibility and cost-sharing arrangements across Canada
While all provinces agree on the need for drug coverage for families receiving social assistance, policies differ with respect to coverage for low-income families who fail to quality for social assistance. The definition of poverty - and, hence, eligibility - differs among provinces, creating regional disparities. This diversity is exacerbated by the variation in cost-sharing requirements. Premiums, deductibles and co-payments, or a combination thereof, are found in all provincial drug plans except for that of Newfoundland. These out-of-pocket costs constitute a user fee required to gain access to necessary medications. In some cases, such fees can present a formidable financial barrier. Furthermore, the application forms and bureaucratic processes associated with some programs require time and a high degree of literacy, including computer and Internet skills. These barriers are greater for new immigrants and for persons who do not speak English or French as first languages.
Individuals working part-time or those in low-wage occupations (the "working poor") are more likely to be either uninsured or without adequate coverage (Health Canada 2000), putting their children at risk of not getting the medications they need. This risk is intensified when one considers that children from poor families have an increased risk of developing health problems and, thus, have greater medication needs (Finkelstein et al. 2002; Wood et al. 2002). The lack of child-specific programs across Canada is troubling. A major disincentive to leaving social assistance is the loss of healthcare benefits. Only Prince Edward Island, Saskatchewan, Alberta and Yukon have programs to ensure that children of the working poor or people leaving social assistance have drug coverage. These "safety net" programs are part of the federal National Child Health Benefit (NCHB) program. Because the provinces have discretion regarding how to spend this money, variation occurs in drug plan policies. Some provinces choose to provide cash handouts directly to families instead of providing drug coverage. In the absence of a federal requirement to spend these monies on pharmaceutical benefits, in provinces that provide cash in lieu of benefits parents may choose to spend the funds on items other than medications and, thus, there is no guarantee that their children will have adequate access to necessary medications.
As with low-income families, public drug coverage for children with serious chronic diseases varies greatly across Canada. While there is consistency in coverage for cystic fibrosis and growth deficiencies, the various programs differ widely in covering other chronic diseases. Unlike other provincial programs, the "umbrella" and disease-specific programs usually have only clinical criteria as their eligibility requirements. Inherent in this principle is the recognition that these children have great medical needs. For these children, ensuring access supersedes considerations of income. However, for the majority of children for whom medications are medically necessary, family income limitations and cost-sharing remain barriers.
Interregional variation in public health insurance plans is also evident in other countries, including the United States. In the 1990s, a significant lack of public healthcare coverage for medications and health services for children became apparent. In 1997, the US federal government introduced the State Children's Health Insurance Program (SCHIP) to provide federal funding to those states wishing to expand coverage for children. Under the SCHIP, the US federal government provides funds to match state contributions up to US$4 billion annually. Funds are used to establish or expand health insurance programs for uninsured children aged up to 19 years who belong to families with incomes that are less than 200% of the federal poverty level. The interest in SCHIP has been strong, with most states applying for federal funds, and health coverage for children appreciably expanded. As a result of this program, the proportion of adolescents from poor families who were uninsured declined by 8% between 1995 to 2002 (Newacheck et al. 2004). An incentive program for federally matching funds to expand medication benefits to children and low-income families should be considered in Canada. We may also look to programs that exist in Scandinavia or other countries that are ranked highly by the Organization for Economic Cooperation and Development because of the existence of health provisions for low-income families with children.
Listing of paediatric-labelled medications
A low proportion of drugs added to provincial formularies in 2003 is approved for use in children, and an even lower one is indicated for paediatric conditions. This situation may result from a low uptake of paediatric-labelled products because children's diseases are not a priority for provincial formulary committees. Alternatively, a low volume of listing may be due to a dearth of products available to treat children's conditions. Children's health may be a low priority to drug manufacturers because they constitute a small fraction of market share and because of concerns regarding the testing of prescription drugs in children. Lack of research and development for products for children's health will result in a low frequency of drugs approved by Health Canada's Therapeutic Products Directorate for use in children.
A lack of availability of paediatric products has led to physicians prescribing adult medications for off-label use in children. Wider clinical testing of new pharmaceutical products in children would provide much-needed efficacy and safety data to permit greater choices for practitioners and allow broader listing decisions. In 1999, as part of an overall program aimed to promote paediatric clinical research, the US Food and Drug Administration's Modernization Act required manufacturers to conduct clinical trials on any medication that was expected to be widely used in children. In exchange for the paediatric clinical data, the FDA provided manufacturers with a six-month extension on their medication patents. This program has been highly successful in stimulating paediatric clinical research - so much so that in 2005, clofarabine was approved for treatment of relapsed or refractory paediatric acute lymphoblastic anaemia. This marked the first time in decades that a novel anti-cancer drug was approved in the United States for use in children before an adult indication was developed (St. Jude Children's Research Hospital 2005). In recent years, the European Union has also moved towards creating incentives for expanded development of medications for children (Commission of the European Communities 2004). Given the multinational character of the pharmaceutical industry, with strong bases in the United States and Europe, it is expected that more paediatric-labelled drugs will be approved for use in Canada and will be considered for provincial formulary listing.
Why do public drug plans vary across Canada?
Why do public drug plans vary so much with respect to eligibility, cost-sharing arrangements and listing decisions across Canada? First, because they can. The 1964 Hall Commission recommended that prescription medications be included as an insured benefit in a universal healthcare program (Ontario Ministry of Health 1990). Despite this recommendation, except for inpatient care, this essential component of healthcare has been consistently omitted from legislation defining the scope of public healthcare coverage and the requirements of universality and portability.
Provinces are free to make their own decisions regarding "who," "what" and "how much." As a result, pharmaceutical policy decisions are influenced by population demographics, as well as political, fiscal, legal and ethical concerns (Rabinovitch 2004). Provinces differ in size as well as demographic make-up. Eastern Canada has proportionally more seniors compared to the Western provinces. Aboriginal people suffer from certain diseases, such as diabetes and infectious disease, at higher rates than non-Aboriginals. Maritime provinces have more unemployment and poverty than other provinces. To a certain extent, pharmaceutical policies reflect these differences. In addition, each province has a fixed budget with which to allocate healthcare resources. As the population size and tax base varies, so do healthcare budgets. Depending on the governing party, the allocation priorities of the provincial governments may differ greatly. Some administrations are more receptive to lobby groups, few of which represent the interests of children. Legal decisions also play a role in what provinces will pay for. The definition of "medically necessary" continues to be a source of contention, particularly with regard to treatments for rare childhood disorders or diseases that require expensive medications, as seen in Tables 3 and 4. When it comes to provincial budget allocation, a utilitarian view - achieving the greatest quantity of health benefits for the most number of people - sometimes prevails over a more compassionate approach that sees to the needs of society's most vulnerable.
What should a public drug plan for children include?
Despite numerous studies chronicling the wide disparities in (adult) public drug programs in Canada (Canadian Institute for Health Information 2004; Health Canada 2000; Jacobs and Bachynsky 2000; Grootendorst 2002; Narine and Sen 1997; Currie and Nielson 1999; Willison et al. 1998; Morgan 2004; Anis 2000), surprisingly little attention has been paid to what would constitute a fair and equitable program providing affordable access to necessary medications. Morgan and Willison (2004) have proposed a national program that would combine last-dollar coverage (benefits commence after a high deductible is reached) with first-dollar coverage for low-income families and other vulnerable segments of the population. This is a good first step. However, more thought needs to go into the "who," "what" and "how much" questions that specifically apply to vulnerable populations. The healthcare needs of children are vastly different than those of adults (Ungar et al. 2003). Whereas a large proportion of adults can be managed by medications for cardiovascular disease, cancer and diabetes, children suffer from a wider variety but less prevalent array of chronic conditions (Smith 1998). These conditions are often age dependent, such that children's medication needs change as they grow and develop.
A number of limitations were present in this study. Because only 2003 formulary updates were examined rather than a fixed basket of products, it is possible that some of the drugs added in one province in 2003 were added previously or subsequently in other provinces. It was therefore not possible to determine whether the variation in new listings among provinces was a result of different rejection rates by provincial decision-makers, differences in the timing of listing decisions or different submission rates by drug manufacturers. A study extending over several years or examining both new and existing listings would clarify this issue. It was also observed that the specific generic versions of drugs and dosage forms sometimes varied by province. This finding may relate to specific purchasing agreements between generic manufacturers and provincial bodies.
The findings presented provide a descriptive first look at interprovincial variation. Future studies are required to examine coverage of drugs deemed essential for children and to look at how each province addresses issues of efficacy, cost-effectiveness, patient adherence and disease management and education.
Conclusion
Drug coverage and drug programs for children vary widely across Canada. Provincial disparities in "who," "what" and "how much" create access barriers to proper healthcare. Both the Romanow and Kirby reports recommended that drug coverage be extended to all Canadians who need it (Romanow 2002; Kirby 2002). The Romanow Commission recommended the establishment of a national formulary to eliminate the disparities in drug benefits across Canada. The creation of the Common Drug Review (CDR) is a first step to achieve this. Although provinces still make the final decisions regarding listing status of each product, by providing a centralized review mechanism the CDR Directorate increases the probability of common listing decisions. Another key recommendation of the Romanow Commission was the establishment of a Catastrophic Drug Transfer, in which the federal government would transfer money to provinces to reduce or eliminate high deductibles and other forms of cost-sharing. The provinces have responded in unison to this suggestion with calls for a federally funded national pharmacare program. And so the debate continues.
The great variation in drug coverage found in this study highlights the need for policy changes. All children across Canada, no matter what province they call home, are entitled to ready and affordable access to the same, comprehensive formulary of medications.
Régimes publics d'assurance-médicaments
pour les enfants au Canada :
Un portrait aux trop nombreuses facettes
Résumé
Historique : Bien que le débat au sujet de l'assurance-médicaments au Canada se poursuive, on accorde peu d'attention à la question des régimes adéquats d'assurance-médicaments pour les populations vulnérables, dont les enfants. L'objectif premier de cette étude consistait à déterminer l'ampleur de la couverture accordée aux enfants par les programmes publics d'assurance-médicaments dans chaque province canadienne.
Méthodes : On a recueilli des données relatives aux régimes d'assurance-médicaments des provinces, des territoires et du gouvernement fédéral et on a obtenu des formulaires actualisés pour 2003. Un modèle de simulation a été établi en vue de montrer les coûts qu'une famille à faible revenu ayant un enfant asthmatique doit payer dans chaque province. Les régimes ont été comparés de façon descriptive. On a résumé statistiquement l'étendue de la variation interprovinciale dans les formulaires approuvés en 2003.
Résultats : En ce qui a trait à l'accès aux médicaments pour les enfants, les régimes provinciaux d'assurance-médicaments varient considérablement quant aux personnes et aux médicaments couverts ainsi qu'au montant que les assurés doivent débourser. On a noté une variation de 39 % entre les provinces dans les formulaires approuvés en 2003 (chi carré p < 0,0001) et une variation de 48 % dans le cas des produits pédiatriques (chi carré p < 0,0001). Dans tout le Canada, seulement 8 % des formulaires approuvés en 2003 portaient essentiellement sur les affections pédiatriques. Dans le modèle de simulation, les coûts correspondaient à 3 % ou moins du revenu familial dans les provinces qui disposent de régimes à l'intention des familles à faible revenu, de régimes de couverture des coûts catastrophiques (Ontario) ou de régimes pour toute la population. Les familles que ne sont pas admissibles aux régimes pour familles à faible revenu ou qui habitent au Nouveau-Brunswick ou à Terre-Neuve devaient payer des frais pouvant atteindre 7% du revenu familial.
Conclusion : En ce qui concerne les avantages accordés aux enfants en matière de médicaments, les régimes d'assurance-maladie provinciaux varient considérablement quant aux bénéficiaires et aux médicaments couverts ainsi qu'au déboursé. Comparativement aux bénéficiaires de prestations aux aînés ou de l'aide sociale, les enfants ne reçoivent aucune couverture complète et uniforme dans toutes les provinces. Bon nombre d'enfants canadiens n'ont donc pas accès aux médicaments dont ils ont besoin en raison de contraintes financières.
About the Author(s)
Wendy J. Ungar, MSC, PHD
Scientist, Population Health Sciences, The Hospital for Sick Children
Assistant Professor, Department of Health Policy, Management and Evaluation,
University of Toronto
Adjunct Scientist, Institute for Clinical Evaluative Sciences
Toronto, ON
Scientifique, Sciences de la santé de la population, Hôpital pour enfants
Professeure adjointe, Département des politiques,
de la gestion et de l'évaluation de la santé, Université de Toronto
Scientifique adjointe, Institute for Clinical Evaluative Sciences
Toronto, ON
Maciej Witkos, MSC
Graduate Student, Department of Health Policy, Management and Evaluation
University of Toronto
Étudiant de troisième cycle, Département des politiques,
de la gestion et de l'évaluation de la santé
Université de Toronto
Acknowledgment
The assistance of the drug plan managers in each province and territory who contributed information to this study is gratefully acknowledged. The authors thank Ms. Magdalena Krstevska for technical assistance and Ms. Cara Mirabelli and Ms. Martha Cousins for useful comments.This research was funded by an operating grant from the Canadian Institutes of Health Research (Grant #37919). Dr. Ungar is supported by a Canadian Institutes of Health Research New Investigator Career award.
Correspondence and reprint requests may be directed to Dr. Wendy Ungar, Population Health Sciences, The Hospital for Sick Children, 555 University Avenue, Toronto, Ontario, Canada M5G 1X8; tel: 416-813-8519; fax: 416-813-5979; email: wendy.ungar@sickkids.ca
References
Anis, A.H. 2000. "Pharmaceutical Policies in Canada: Another Example of Federal-Provincial Discord." Canadian Medical Association Journal 162: 523-26.
Canadian Association for Pharmacy Distribution Management. 2004. 2004 Guidebook on Government Prescription Drug Reimbursement Plans and Related Programs. Woodbridge, ON: Author.
Canadian Institute for Health Information. 2004. Drug Expenditures in Canada 1985-2003. Retrieved July 31, 2005. < http://secure.cihi.ca/cihiweb/dispPage.jsp?cw_page=
PG_283_E&cw_topic=283&cw_rel=AR_80_E#full >.
Canadian Pharmacists Association. 2003. Compendium of Pharmaceuticals and Specialties. The Canadian Drug Reference for Health Professionals. Toronto: Author.
Canadian Pharmacists Association. 2004 (December). Provincial Drug Benefit Programs. A Summary of Prescription Drug Benefit Plans Sponsored Provincially in Canada (28th ed.). Toronto: Author.
Commission of the European Communities. 2004. Proposal for a Regulation of the European Parliament and of the Council on Medicinal Products for Paediatric Use and Amending Regulation (EEC) No. 1768/92, Directive 2001/83/EC and Regulation (EC) No. 726/2004. Retrieved July 31, 2005. < http://dg3.eudra.org/F2/Paediatrics/
docs/_2004_09/EN.pdf >.
Currie, G.R. and N.L. Nielson. 1999. "Models for Funding Prescription Drug Programs: Institute of Health Economics." Working Paper 99-5.
Ernst, P., J.M. Fitzgerald and S. Spier. 1996. "Canadian Asthma Consensus Conference: Summary of Recommendations." Canadian Respiratory Journal 3(2): 89-100.
Finkelstein, J.A., P. Lozano, H.J. Farber, I. Miroshnik and T.A. Lieu. 2002. "Underuse of Controller Medications among Medicaid-Insured Children with Asthma." Archives of Pediatric and Adolescent Medicine 156(6): 562-67.
Grootendorst, P. 2002. "Beneficiary Cost Sharing under Provincial Prescription Drug Benefit Programs: History and Assessment." Canadian Journal of Clinical Pharmacology 9(2): 79-99.
Health Canada. 1997. National Forum on Health Final Report - Volume I. Canada Health Action: Building on the Legacy. Chapter 1.2: "Building a More Integrated System." Retrieved July 31, 2005. < http://www.hc-sc.gc.ca/english/care/health_forum/
publications/finvol1/index.html >.
Health Canada. 2000 (March). Canadians' Access to Insurance for Prescription Medicines. Ottawa: Applied Management in association with Fraser Group Tristat Resources.
Health Canada. 2001. Health Policy and Communications Branch. Health Expenditures in Canada by Age and Sex, 1980-81 to 2000-01. Catalogue No. H21-172/2001. Ottawa: Minister of Public Works and Government Services Canada.
Health Canada. 2004. Drug Product Database (DPD). Retrieved July 31, 2005. < http://www.hc-sc.gc.ca/hpb/drugs-dpd >.
Jacobs, P. and J. Bachynsky. 2000. "Public Policies Related to Drug Formularies in Canada: Economic Issues." Edmonton: Institute of Health Economics. Working Paper 00-2: 1-51.
Kirby, M. 2002 (October). The Health of Canadians - The Federal Role. The Standing Senate Committee on Social Affairs, Science and Technology (Chair: The Honourable Michael J.L. Kirby, Deputy Chair: The Honourable Marjory LeBreton). Retrieved July 31, 2005. < http://www.parl.gc.ca/37/2/parlbus/commbus/senate/
com-e/SOCI-E/rep-e/repoct02vol6-e.htm#TABLE%20
OF%20CONTENTS >.
Kozyrskyj, A.L., C.A. Mustard, M. Cheang and F.E.R. Simons. 2001. "Income-Based Drug Benefit Policy: Impact on Inhaled Corticosteroid Use in Manitoba Children with Asthma." Canadian Medical Association Journal 165: 897-902.
Manitoba Health. 2004. Manitoba Drug Benefits and Interchangeability Formulary. Retrieved July 31, 2005. < http://www.gov.mb.ca/health/mdbif >.
Mannino, D.M., D.M. Homa, C.A. Pertowski, A. Ashizawa, L.L. Nixon, C.A. Johnson et al. 1998 (April 24). "Surveillance for Asthma - United States, 1960-1995." US Centers for Disease Control, Morbidity and Mortality Weekly Report Surveillance Summary 47(SS-1): 1-28.Millar, W.J. 1998. "Childhood Asthma." Health Reports 10(3): 9-21. Statistics Canada Catalogue No. 82-003.
Morgan, S. 2004. "Sources of Variation in Provincial Drug Spending." Canadian Medical Association Journal 170(3): 329-30.
Morgan, S.G. and D.J. Willison. 2004. "Post-Romanow. Pharmacare: Last-Dollar First... First-Dollar Lost?" Healthcare Papers 4(3): 10-20.
Narine, L. and M. Sen. 1997 (November). Pharmaceutical Cost Containment Policies: Intended and Unintended Impacts. Toronto: Faculty of Medicine, University of Toronto.
Newacheck, P.W., M.J. Park, C.D. Brindis, M. Biehl and C.E. Irwin, Jr. 2004. "Trends in Private and Public Health Insurance for Adolescents." Journal of the American Medical Association 291(10): 1231-37.
Ontario Ministry of Health. 1990. Prescriptions for Health: Report of the Pharmaceutical Inquiry of Ontario. Toronto: Author.
Rabinovitch, S.R. 2004. Evaluation of the Romanow Commission's Recommendation for a National Prescription Drug Formulary. University of Toronto Faculty of Law. Health Law & Policy Group Student Working Paper. Retrieved July 31, 2005. < http://www.law.utoronto.ca/healthlaw >.
Romanow, R. 2002 (November). Commission on the Future of Healthcare in Canada: Building on Values - The Future of Healthcare in Canada. Final Report. Retrieved July 31, 2005. < http://www.hc-sc.gc.ca/english/care/
romanow/hcc0086.html >.
Skidmore-Roth, L. 2003. Mosby's Drug Guide for Nurses. Toronto: Elsevier Science.
Smith, P.S. 1998. "Determining Whether Managed Care Formularies Meet the Needs of Pediatric Patients." American Journal of Managed Care 4(9): 1328-35.
St. Jude Children's Research Hospital. 2005. "Giving Children Access to Drug Clinical Trials is Crucial, Clofarabine Approval Shows." Retrieved July 31, 2005. < http://www.eurekalert.org/pub_releases/
2005-05/sjcr-gca050205.php >.
Ungar, W.J., C. Daniels, T. McNeill and M. Seyed. 2003. "Children in Need of Pharmacare - Medication Funding Requests at the Toronto Hospital for Sick Children." Canadian Journal of Public Health 94(2): 121-26.
US National Library of Medicine and National Institutes of Health. Medline Plus Drug Information. Retrieved July 31, 2005. < http://www.nlm.nih.gov/medlineplus/druginformation.html >.
Willison, D., P. Grootendorst and J. Hurley. 1998 (September). Variation in Pharmacare Coverage Across Canada. Centre for Health Economics and Policy Analysis, McMaster University. Working Paper 98-8.
Wood, P.R., L.A. Smith, D. Romero et al. 2002. "Relationships between Welfare Status, Health Insurance Status, and Health and Medical Care among Children with Asthma." American Journal of Public Health 92(9): 1446-52.
Comments
Be the first to comment on this!
Personal Subscriber? Sign In
Note: Please enter a display name. Your email address will not be publically displayed