Healthcare Quarterly
Aiming for Zero Preventable Deaths: Using Death Review to Improve Care and Reduce Harm
Abstract
In 2005, our organization set a goal of zero preventable deaths by 2010 – notionally a sound goal but extremely challenging to measure, monitor and evaluate. The development of an interdisciplinary Death and Adverse Event Review process has provided a measure and framework for action to decrease adverse events (AEs) that cause harm.
Death and Adverse Event Review is a formal process in which trained reviewers consider patient deaths using a modified Global Trigger Tool to establish the presence of AEs or quality of care issues that may have potentially led to death or harm. When identified, these charts go to second-level review by a physician/interdisciplinary team to determine recommendations for actions to prevent future reoccurrences. Data have provided trending of system influences to patient safety. In 2008–2009, 1,817 deaths were reviewed and AE rates of 12.1% and 16.3% were identified. There were 422 AEs and 114 quality of care issues identified for follow-up. Of the 4.7% and 6.3% referred to the physician/interdisciplinary team for secondary review, 2.3% and 2.6% resulted in recommendations for improvement. In addition to local improvements, many system improvements have occurred as a result of the review, such as proposed minimum standards for physician documentation; a formal review of post-operative guidelines for patients with sleep apnea; and a working group to review nursing documentation, communication/follow-up of vital signs, fluid balance and pain management. The Death and Adverse Event Review process provides a new critical level of detail that supports continuous improvements to our care processes and ongoing progress toward our goal of zero preventable deaths.
With the international focus on the measurement of hospital standardized mortality ratios (HSMRs), as well as the alarming frequency of adverse events (AEs) in hospitals, more and more hospitals are seeking strategies to understand the influences within the complexity of healthcare that may directly contribute to patient harm and death. As noted in the Canadian Adverse Events Study, 7.5% of Canadians may experience, as a result of healthcare management, an AE, which includes an unintended injury or complication that can lead to disability, prolonged hospital stay or death; this number rises to 10.9% of those receiving treatment in teaching hospitals (Baker et al. 2004). Measuring the prevention and reduction of AEs is challenging as it is an inherently complex and subjective process. Traditionally, patient safety events are identified and calculated according to voluntary, spontaneous reporting through occurrence reporting systems. However, studies have shown that only 10–20% of occurrences are actually reported and, of those, 90–95% cause no harm (Institute for Healthcare Improvement [IHI] 2009). Studies have also shown that medical record reviews (chart reviews) elicit significantly higher numbers of reports than does voluntary reporting. In a study by Levinson (2010) comparing multiple methods for identifying AEs, a review of medical records by nurses or physicians was found to be an effective way to identify AEs. Another study comparing chart review with occurrence reporting demonstrated that 83% of AEs were identified by chart review, whereas only 7% were identified by occurrence reporting (Baba-Akbari Sari et al. 2006).
As such, hospitals need effective methods to quantify and understand actual AE rates with a critical level of detail that allows for more confident and definitive decision-making. If hospitals do not have an accurate reflection of their true AE trends and rates, much effort can be focused on areas that may be reported frequently but are not an accurate reflection of high-risk areas of harm to patients. Critical analysis of significant harm events can expose actionable root causes versus responses to trended occurrences. This paper outlines the interdisciplinary Death and Adverse Event Review process that has been developed at Hamilton Health Sciences (HHS) to more accurately measure and identify AEs that cause harm, and to provide a framework for action to decrease AEs that cause harm.
Death Review at HHS
HHS is a seven-site, 1,000-bed regional tertiary care facility composed of six hospitals and a cancer centre, and it has approximately 1,500 deaths per year. In 2005, HHS set a goal of zero preventable deaths by 2010. This was notionally a sound goal but practically a goal that was extremely challenging to measure, monitor and evaluate. HHS recognized that to be successful an effective and accurate means of identifying AEs would be required. This vision of zero preventable deaths, while arguably a stretch goal, has proven to be a driving force in engaging staff in the patient safety journey. Leape and Berwick (2005) suggest that with sufficient will and leadership, we can aim for ambitious goals. The obstacles lie in beliefs, intentions, cultures and choices, all of which can change. While some may argue that zero preventable deaths is not truly attainable, this goal is aligned with our philosophy of continuous quality improvement, and truly no other goal would be acceptable to our staff or the patients and families we serve.
Prior to 2007, chart reviews were completed on all deceased patients; however, this process was fraught with challenges. The reviews occurred primarily at the physician level, and learning, communication and resolution of issues resided primarily within a particular department. There was no structured accountability, were few/limited forums for interdisciplinary discussion and was little corporate dissemination and sharing of findings. This reality, coupled with the literature findings, prompted HHS to reassess how it would successfully realize our goal of zero preventable deaths.
The new process has trained patient safety specialist reviewers (PSSRs) reviewing all adult patient deaths at HHS within 48–72 hours of death (whenever possible). The process is outlined in Figure 1. When an AE is found that may have potentially contributed to a patient's death it is referred for second level review by a physician reviewer. If the reviewer is in agreement, the chart is then reviewed by a multidisciplinary team for recommendations and follow-up which are forwarded to appropriate stakeholders. If not already completed, an Occurrence Report is initiated for any AEs in which there was moderate to severe harm noted by the PSSRs during the chart reviews and forwarded for follow-up and investigation. In addition, any "near miss" occurrences or quality of care issues are flagged and forwarded to the respective stakeholder groups.
Methods
The chart review process uses a modified version of the Global Trigger Tool methodology developed by IHI. The IHI methodology involves a retrospective review of patient charts using "triggers" (clues) to identify possible AEs. The triggers are, in essence, clinical indicators thought to be predictive of the presence of an AE and to signal the need for further review of the situation in which they occur. HHS customized the IHI triggers to include screening tool criteria identified in the Canadian Adverse Events Study and criteria identified by HHS physicians and interdisciplinary practice chiefs.
During the review, details of all patient demographics are collected. Occurrences (AEs, near misses or quality of care issues) are collected, quantified and categorized in a manner consistent with our organizational occurrence-reporting classifications and risk levels. This allows for comparisons of data from both sources. Every six months, the data are analyzed to understand trends and progress.
Results
Measurement of an Actual AE Rate
From March 2008 to March 2009, 1,817 deaths were reviewed and AE rates of 12.1% and 16.3% were identified. This represents 422 AEs and 114 quality of care issues that were identified for follow-up. Of the 1,817 deaths reviewed, only 4.7% and 6.3% were referred to the physician/interdisciplinary team for secondary review, of which only 2.3% and 2.6% resulted in recommendations for improvement (Table 1).
| Table 1. Adverse events and referrals for second-level review | ||
| Adverse Events | Review (March 2008–August 2008) | Review (September 2008–March 2009) |
| Number of patient charts reviewed in total | 840 | 977 |
| Number of adverse events found | 188 | 234 |
| Number of patients (deaths) with adverse events | 137 (16.3%) | 118 (12.1%) |
| Number of charts referred to local Death Review Committee for second-level review | 53 (6.3%) | 46 (4.7%) |
| Number of charts accepted by local Death Review Committee | 20 (2.4%) | 26 (2.7%) |
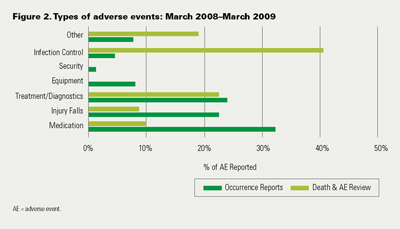
Of interest is the difference in AE trends revealed by the traditional HHS occurrence reporting and the Death and Adverse Event Review process. While occurrence reporting tends to highlight very visible harm (medication error, falls, procedural/treatment errors), the Death and Adverse Event Review process seems to highlight less visible errors (infection control – hospital-acquired infections, procedural/treatment events and miscellaneous events such as self-extubations, aspirations and documentation issues that led to harm). These trends have allowed for focused targeted action to decrease AEs (Figure 2).
Move Your Dot Methodology
The Death and Adverse Event Review process also uses the IHI Move Your Dot strategy, which identifies how organizations might reduce mortality rates and consequently improve HSMR results. Using the model, deaths are categorized into four quadrants, which suggest where initiatives should be focused to impact mortality rates (Table 2). Results indicate that a focus is needed on box D (1,077 deaths), which further suggests that, since the outcome was death, these patients were in fact high risk but possibly not assessed as such. Work here might involve addressing core systems issues such as patient safety and specifically medication safety (IHI 2003). These results have fortuitously aligned with many current HHS initiatives implemented to date and support planning for future initiatives. Some examples of initiatives to date include the transfer of accountability guidelines, automated medication dispensing units and unit dose medication systems, rapid response teams and the communication of critical test results.
| Table 2. HHS Move Your Dot Matrix Data (March 2008–March 2009) | ||
| ICU Admission | No ICU Admission | |
| Comfort care only | "Box A": 50 deaths | "Box B": 190 deaths |
| Not comfort care only | "Box C": 500 deaths | "Box D": 1,077 deaths |
| HHS = Hamilton Health Sciences; ICU = intensive care unit. | ||
Process Evaluation
Recently, a process evaluation was conducted with internally designed surveys with the 18 members of the Death Review Committee, including physicians, profession chiefs and members of the Quality and Patient Safety Team. The survey had a 61% response rate (11 members). As well, 48 stakeholders were also surveyed including clinical managers and directors involved in the process and profession chiefs. There were 22 surveys returned (46%). Key highlights of the evaluation were as follows:
- Overall value of primary screening by reviewers was rated by 82% of respondents as very valuable to having excellent value.
- Death Review Committee members found the new process to be timely and an efficient use of time, and 82% noted the process to be an improvement.
- Impact of the Death and Adverse Event Review process to improving patient safety was rated as good to excellent by 82% of the Death Review Committee respondents and by 67% of the stakeholder respondents.
- One area for improvement was improved sharing of the learnings from the death review; 67% of stakeholders identified this as an opportunity.
Discussion and Implications
To date there have been limited Canadian data available with respect to quantifying AE rates in hospitalized patients. The Canadian Adverse Events Study (Baker et al. 2004) found the overall incidence rate of AEs in patients at teaching hospitals was 10–11%. Other (non-Canadian) studies revealed that 2.3–16.6% of patents in acute care hospitals experienced one or more AEs (Baker et al. 2007). Most recently, an American study (Tolchin et al. 2007) found that 25% of patients who died had experienced an AE that may have contributed to their death. The significant disparity between the calculated/reported AE rates in the literature reflects the inherently complex and subjective nature of AE identification, measurement and reporting. This reality, coupled with the relative newness of patient safety research, makes identifying an expected AE rate or benchmark extremely difficult. Undoubtedly, it is very challenging for clinicians to distinguish the impact of an AE from other causes of poor outcomes, that is, determining whether a causal relationship actually exists between the AE and harm or death. Determining the preventability of AEs is equally difficult. That said, in an effort to identify potential AE rates at HHS, the results of the Canadian Adverse Events Study were extrapolated into "expected rates" for HHS; these and the actual AE rates from the Death and Adverse Event Reviews and are summarized in Table 3. Caution does need to be given to the extrapolation of these results given the differences in study groups (live and deceased patients), sample size and methodology.
| Table 3. Comparison of AE rates in the Canadian Adverse Events Study to Extrapolation of actual AE rates for HHS | |||
| Canadian Adverse Events Study* | Extrapolation of Canadian Adverse Events Study Results to HHS† | Death and Adverse Event Review Results‡ | |
| Number of admitted patients who will have one or more AE | 10.3 per 100 admissions to teaching hospitals | 4,120 admissions per year will experience an AE | 12.3% of all patients who die experience an AE; if deaths could be compared to admission sample = 4,920/year§ |
| Number of admitted patients who will experience one or more AE and die | 1.6 per 100 admissions | 640 admissions per year will experience one or more AE and die | 202 deaths with one or more AE (0.5% of all admissions) |
| AE = adverse event; HHS = Hamilton Health Sciences. | |||
|
*Randomly selected chart reviews (admissions). Data from Baker et al. (2004). |
|||
|
†Based on 40,000 admissions annually. |
|||
|
‡September 2008–March 2009 annualized and assuming 40,000 admissions annually. |
|||
|
§Acuity/complexity of deaths would explain higher rate given not a comparable population. |
|||
AE Trends
The IHI (2009) notes that assessment of patient safety has traditionally relied on monitoring of systems and analyses of single or aggregate events. Continuous systematic monitoring of the frequency and nature of AEs has rarely been performed. This has made it difficult for organizations to know definitively if the care they provide is becoming safer. Tracking AEs over time is a useful way to tell if changes being made are improving the safety of the care processes. The Death and Adverse Events Review enables us to focus on fixing faulty system processes to improve patient safety. As well, the process has focused the organization more on hidden harm system issues that may not have been addressed with the use of only occurrence reporting data.
Physician Engagement
The interdisciplinary approach has allowed for a better understanding of the whole system and the subsequent identification of system issues for improvement. Previously, deaths were reviewed by busy clinicians, something that happened with variable success depending on the department involved and the actual number of deaths combined with the lack of structured accountability. The use of nurse reviewers to screen charts prior to physician review has promoted physician engagement, allowing them to focus on events that have a defined question associated with them. Consequently, physicians are now able to complete the necessary reviews and assume an active role in improving system influences to patient safety.
There has been a notable increase in the number of charts that the physicians conducting second-level review have "accepted" from the PSSRs. In the first six-month period of review, only 15.4% of charts were accepted versus 57% of charts in the third review period. While there have been some minor modifications to the trigger tools and communication processes, the increase can in part be attributed to growing medical support for the process and the increasing expertise of the PSSRs. The culture of death review has also demonstrated significant change over the past year. The focus on interdisciplinary review, open discussion and challenging colleagues to make recommendations to ensure events do not happen again has become increasingly apparent, as have the refinement and attention to action.
Improved Death Review Processes
Many corporate and local level changes and initiatives have resulted from the Death and Adverse Event Review process. From a process perspective, all deaths at HHS are now reviewed within 72 hours whenever possible – a considerable and significant improvement from the previous experience of months required to complete some reviews. The use of a PSSR role and the application of trigger tools have considerably reduced the number of charts that are reviewed by physicians and the multidisciplinary teams. Only 4.7% and 6.3% of all deaths are forwarded by PSSRs for second-level review, thus allowing clinicians to focus on those charts with suspected issues versus a review of every patient death. In addition, there is continued evolution of the review processes: there is a growing interest in creating multidisciplinary teams for death review and a gradual shift away from reviews by the primary physicians.
Improved Processes of Care
In addition to process improvements, many local and organizational improvement initiatives have resulted from the Death and Adverse Event Review process; a sample of some of these follow:
- A corporate Back to Basics initiative group has been formed to address an ongoing lack of documentation of fluid balance, weight and vital signs and/or critical follow-up of abnormal results to help prevent late rescues of patients.
- There has been a joint recommendation from the surgery and medicine departments to the Medical Advisory Committee to develop clear, basic minimum standards for physician documentation to better enable teams to follow consistent plans of care.
- There has been a recommendation for the chiefs of surgery and anesthesia to formally review the current guidelines for the monitoring and identification of patients with sleep apnea to allow for the identification of at-risk patients and appropriate planning for care.
- A hospital committee has been established to review the care of patients with a history of drug abuse who require intravenous or central lines to prevent harm resulting from self-injections.
- A "transitional" transfer of accountability protocol for nurses was developed to make sure that appropriate and critical information is communicated when patients are transported "off units"; this will ensure that all areas are aware of risk issues for patients during transitional periods.
In addition, an extensive number of local level initiatives have been implemented following the review of referred cases.
Challenges and Lessons Learned
To date, significant progress has been made to refine and improve the Death and Adverse Event Review process. With this implementation, have come lessons in physician engagement and sustainability.
Physician Engagement
Significant engagement and support by key physician leaders including the vice-president of medicine and the Medical Advisory Committee (MAC) chair was essential to communicate the need for change and to assist with communication and support for process changes. Consistent attendance by the MAC chair to the HHS Death Review meetings was also instrumental in the ongoing development of a learning culture that is focused on opportunities to improve safety and clearly identified actions.
Sustainability
To ensure a sustainable and continually evolving process, multiple strategies were needed, including the following:
- Dedicated reviewers with significant training to continually improve and sustain the initiative
- An identified manager to provide oversight and operational management of the process
- Integrated inter- and intra-reviewer reliability auditing
- Clear accountability and reporting framework established with reports to the corporate Death Review Committee, corporate Quality Patient Safety Steering Committee and the MAC
- Continuous refinement of the process including a stakeholder evaluation completed in June 2009
- Integration of process with risk management practices
- Program- and unit-specific results presented biannually to management teams to share with front-line staff
- Integration of the trends and data into the quality and patient safety three-year plan
Conclusion
The purpose of the Death and Adverse Event Review process is to provide the detail required to lead to system-level improvements and to accelerate HHS to zero preventable harm and deaths. While the process has undoubtedly been challenging and complex, significant improvements and understanding of system issues have been gained as a result of its implementation. While many US hospitals employ a similar process or iteration, there are few Canadian hospitals that have adopted such a review process. To that end, HHS has relied on literature and internal expertise to guide our efforts; while there are a myriad of equally compelling and important initiatives that require attention and resources, HHS is committed to the Death and Adverse Event Review process. To sustain the current progress, we continue to refine the process. As confidence is gained with the identification of trends and system issues that are contributing to AEs and potentially death, this process may evolve to a sampling approach. This would, in turn, create an opportunity to use resources to review patient populations at HHS other than the deceased. We believe we will continue to learn from the reviews and that the process is just one of many important tools that we are utilizing to understand patient safety issues at HHS. We are pleased that our efforts to date have resulted in many improvements to our care delivery system – improvements that will ensure that HHS delivers on its mission to be "leaders in exemplary care, innovation and academic excellence."
About the Author(s)
Rosanne Zimmerman, RN, BHScN, MEd, is a manager of patient safety and clinical resource management at Hamilton Health Sciences, in Hamilton, Ontario.
Sharon Pierson, RN, BScN, MPA, is the director of quality, patient safety & clinical resource management at Hamilton Health Sciences.
Richard McLean, MD, FRCP(C), is the vice-president of medical affairs and quality at Hamilton Health Sciences.
Sue Anne McAlpine, RN, BScN, GNC(C), is a patient safety specialist at Hamilton Health Sciences.
Carole Caron, RN, BEd, MA(c), is a patient safety specialist at Hamilton Health Sciences.
Beth Morris, RN, BSN, MS, is a patient safety specialist at Hamilton Health Sciences.
Janie Lucas, RN, BScN, MSc, is a quality specialist at Hamilton Health Sciences.
References
Baba-Akbari Sari, A., T. Sheldon, A. Carcknell and A. Turnbull. 2006. "Sensitivity of Routine System for Reporting Patient Safety Incidents in an NHS Hospital: Retrospective Patient Case Note Review." BMJ 10: 1136–40.
Baker, G.R., L. Jeffs and M.P. Law. 2007. "Improving the Safety and Quality of Health Care in Canada." In N. MacKinnon, ed., Safe and Effective: The Eight Essential Elements of an Optimal Medication Use System. Ottawa, ON: Canadian Pharmacists Association.
Baker, G.R., P.G. Norton, V. Flintoft, R. Blais, A. Brown, J. Cox et al. 2004. "The Canadian Adverse Events Study: The Incidence of Adverse Events among Hospital Patients in Canada." Canadian Medical Association Journal 170 (11): 1678–86.
Griffin, F.A. and R.K. Resar. 2009. IHI Global Trigger Tool for Measuring Adverse Events (2nd ed.; IHI Innovation Series White Paper). Cambridge, MA: Institute for Healthcare Improvement.
Institute for Healthcare Improvement. 2003. Move Your Dot: Measuring, Evaluating, and Reducing Hospital Mortality Rates (Part 1; IHI Innovation Series White Paper). Boston: Institute for Healthcare Improvement.
Leape, L.L. and D.M. Berwick. 2005. "Five Years after to Err Is Human: What Have We Learned?" Journal of American Medical Association 293(19): 2384–90.
Levinson, D. 2010. Adverse Events in Hospitals: Methods for Identifying Events. Washington, DC: United States Department of Health and Human Services, Office of Inspector General.
Tolchin, S., R. Brush, P. Lange, P. Bates and J. Garbo. 2007. "Eliminating Preventable Death at Ascension Health." Joint Commission Journal on Quality and Patient Safety 33(3): 145–54.
Comments
Be the first to comment on this!
Personal Subscriber? Sign In
Note: Please enter a display name. Your email address will not be publically displayed